More Information
Submitted: February 27, 2026 | Accepted: March 26, 2026 | Published: March 27, 2026
Citation: Parenago OP. Ways of Synthesis of Applied Catalysts Using Supercritical Media. Ann Adv Chem. 2026; 10(1): 033-037. Available from:
https://dx.doi.org/10.29328/journal.aac.1001062
DOI: 10.29328/journal.aac.1001062
Copyright license: © 2026 Parenago OP. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestriCted use, distribution, and reproduCtion in any medium, provided the original work is properly cited.
Keywords: Supercritical carbon dioxide; Heterogeneous catalysis; Nanocatalysts; Metal-containing catalysts; Catalyst immobilization; Laser ablation
Ways of Synthesis of Applied Catalysts Using Supercritical Media
Parenago OP*
Institute of Petrochemical Synthesis of the Russian Academy of Sciences, Moscow, Russia
*Address for Correspondence: Parenago OP, Institute of Petrochemical Synthesis of the Russian Academy of Sciences, Moscow, Russia, Email: [email protected]
We conducted a multivariable logistic regression analysis to identify factors that predicted iron supplementation for 90 days or longer among pregnant women in Zambia. The analysis included 7,356 weighted observations. The results showed that the model was statistically significant (Wald X² = 149.27, p < 0.001). This means that the predictors played a crucial role in forecasting adherence to iron supplementation. Education was the most critical factor in whether people followed their iron supplement regimen. Pregnant women with higher education levels were twice as likely (AOR = 2.04, 95% CI: 1.45-2.88, p < 0.001) to comply with iron supplementation for 90 days or more compared to those without education. Pregnant women living in urban areas also predicted adherence to iron supplementation (AOR = 1.25, p = 0.021). There were significant regional differences. Pregnant women in the Eastern and Southern regions were more likely to adhere to iron supplementation compared to those in Lusaka. However, age did not emerge as a significant predictor of iron supplementation when we considered other factors. The model explained nearly 11% of the variation in iron supplementation (Pseudo R² = 0.11).
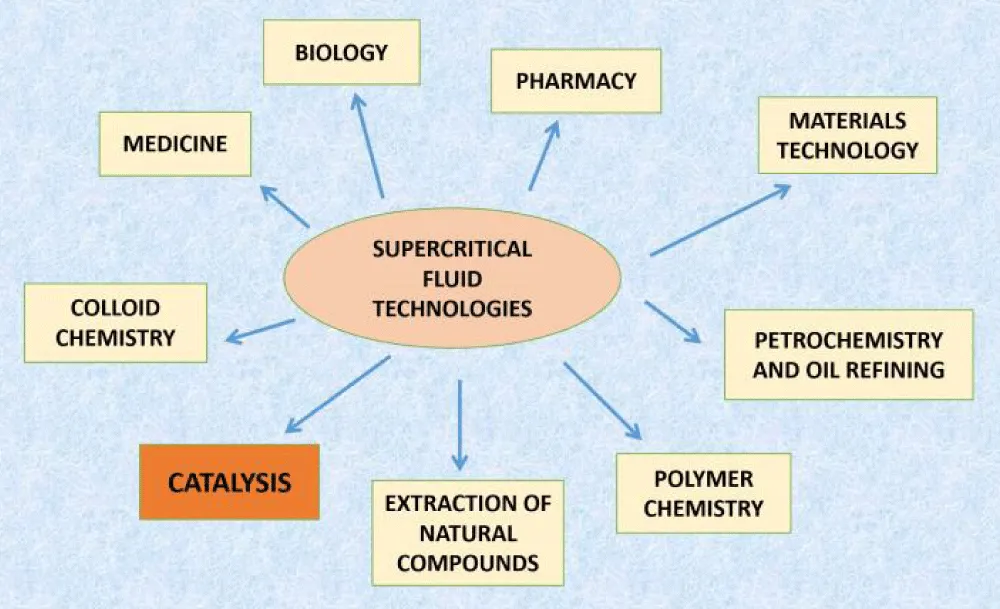
In recent years, there has been a significant increase in research and development of supercritical technology and its practical application in various fields of chemistry and other areas (Figure 1). Research in the field of petroleum chemistry is particularly valuable, including work in the areas of petrochemicals, natural gas chemistry, oil refining processes, and the production of pure chemical products.
Figure 1: Areas where supercritical technologies are used.
Now we are moving on to the main application of supercritical technologies, which is catalysis. This process promises us many interesting developments. Firstly, it allows for the synthesis of pure and highly efficient organic compounds, including the production of high-molecular-weight polymers. Secondly, catalysis offers the opportunity to synthesize various metal-containing systems, either organic or inorganic, which act as catalysts in various organic reactions.
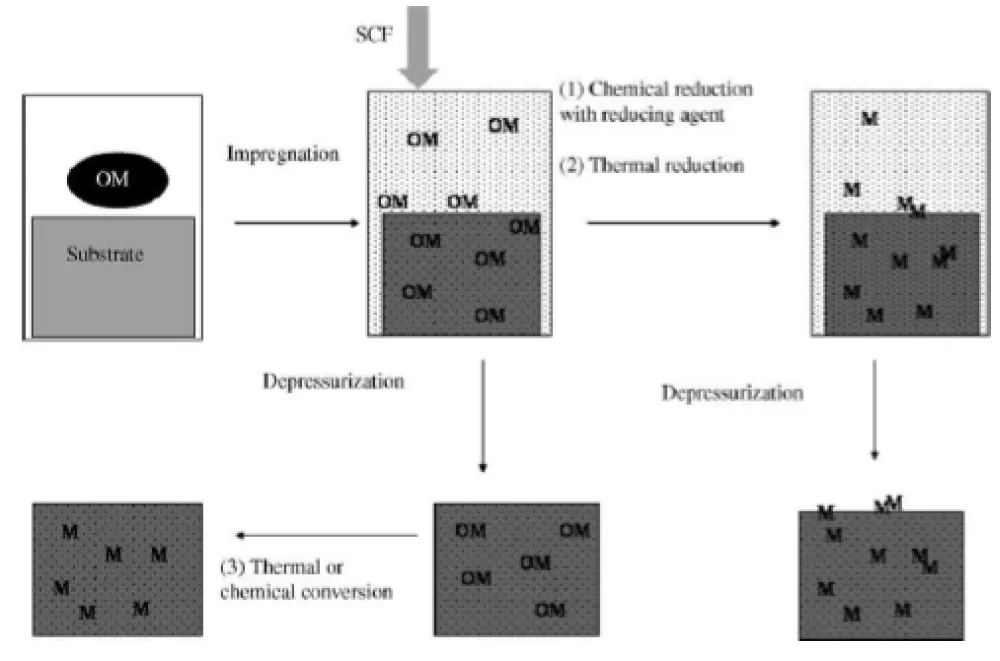
We are currently presenting a review [1] that discusses the synthesis of heterogeneous catalysts using supercritical carbon dioxide technology (Figure 2). The method for impregnating metal compounds on a carrier is as follows. The metal compound is placed in a chamber and treated with supercritical carbon dioxide at a temperature of 31°C and a pressure of 73,8 MPa. After impregnation of OM in the pores of carriers the metal compound is then reduced using hydrogen gas or another reducing agent, and it can be heated to form metal compounds. The pressure and temperature are then reduced, and the metal is deposited on the carrier. Finally, after the depressurization process, the mixture is poured into a separate chamber for final use.
Figure 2: The method for impregnation of OM (metal containing compounds).
The proposed method is highly efficient, and it also has a significant impact on human health and the environment. Unlike strong and hazardous solvents commonly used in the synthesis of many catalytic systems, carbon dioxide is a low-risk option. Carbon dioxide is non-toxic, non-flammable, and non-explosive.
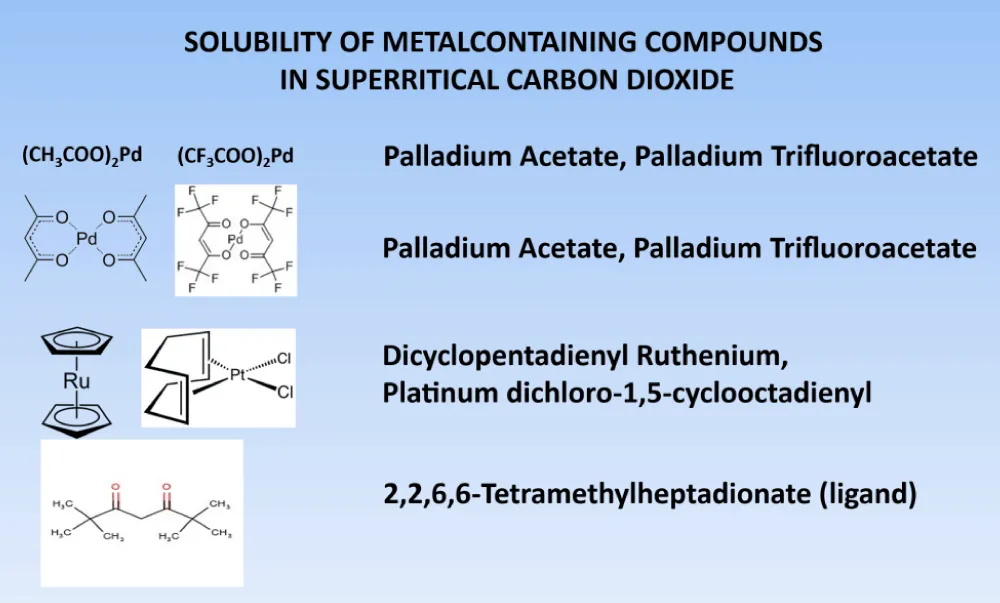
In many ways, this method works well when the solubility of metal complexes in supercritical carbon dioxide is high enough. These data have been published in several reviews [2-4]. Some data are shown in Figure 3.
Figure 3: There are some of the metals with different ligands.
Compounds such as palladium triacetates dissolve very well in supercritical carbon dioxide, and fluorine-containing metal complexes dissolve very well. 2,2,6,6-tetramethylhaptodione for metal complexes are good ligands. Inorganic or polymeric carriers are also very important for the creation of deposited metal-organic catalysts. For example, oxide are good carriers for its: Al2O3, SiO2, TiO2, Fe2O3, ZiO2, mesoporous carriers, such as: MCM-41 or SBA-15. Mesoporous carriers (MCM-41, SBA-15) they are also use. In addition to them, polymer carriers are also used: poly-α-olefins, polypropylene imine dendrimers, super crosslinked styrene and others.
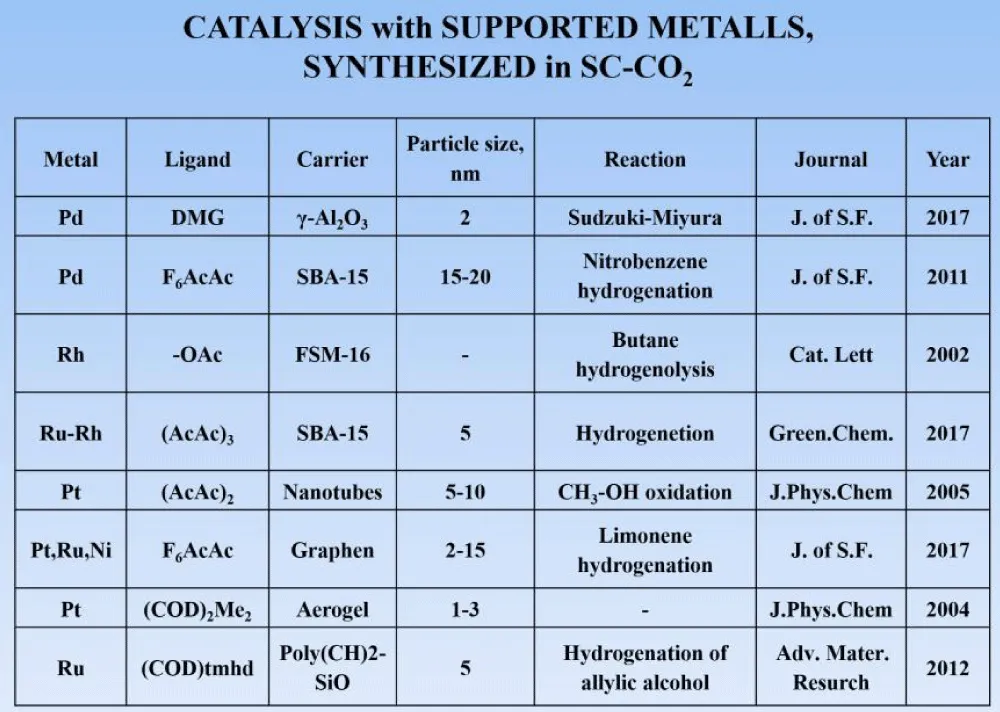
In the table (Figure 4), we demonstrate you a list of some reactions that were carried out by a number of foreign researchers on the synthesis of catalysts using supercritical carbon dioxide. It can be seen that by using various platinum group metals and their ligands as catalysts, as well as various carriers, the resulting contacts had a size of the order of nanometers. The reactions used were the hydrogenation of various compounds, their hydrogenolysis, and oxidation reactions.
Figure 4: A series of works completed in recent years.
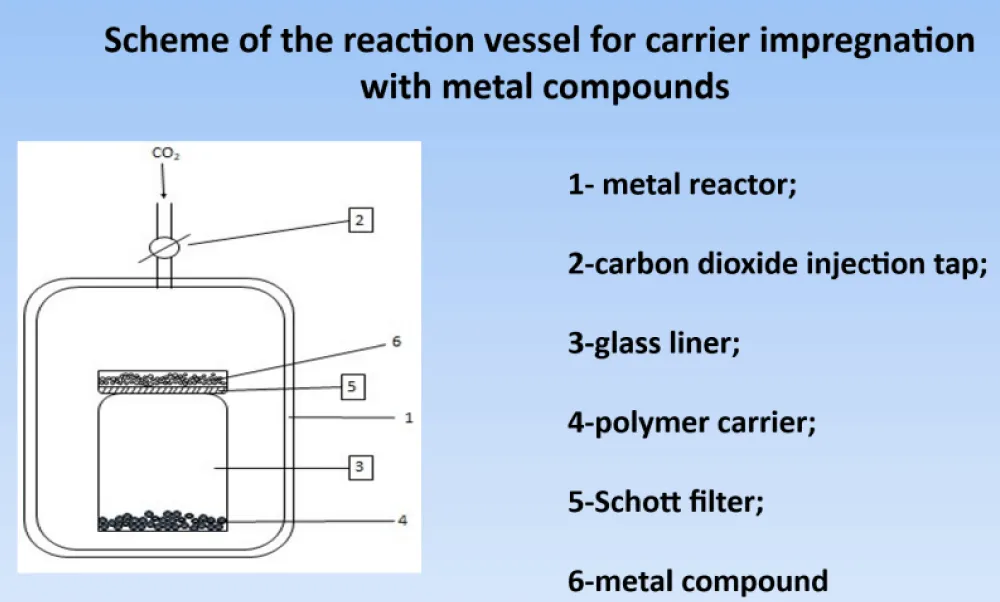
We are now presenting a diagram of a metal reactor for the introduction of supercritical carbon dioxide, which contains a glass volume (100 ml) for the solution of a metal component that will interact with the carrier and form a supported metal-organic compoud on the carrier (Figure 5).
Figure 5: The scheme of the reactor for impragnation of metal to the carrier.
The conditions under which the impregnation of metals on the carrier and the reduction of metall were carried out are listed below. The impragnation was the next: the ratio of MC/PC = 5-40/50-100 mg/mg, where MC = metall conttaining/compounds, PC = impgnation of polymer carriers. P(CO2) = 15 MPa, T,o = 40-50, duration 0,5-24 h. Reduction P(H2) = 5,0-6,5 MPa, T,oC = 50-120, duration =4 -10 h.
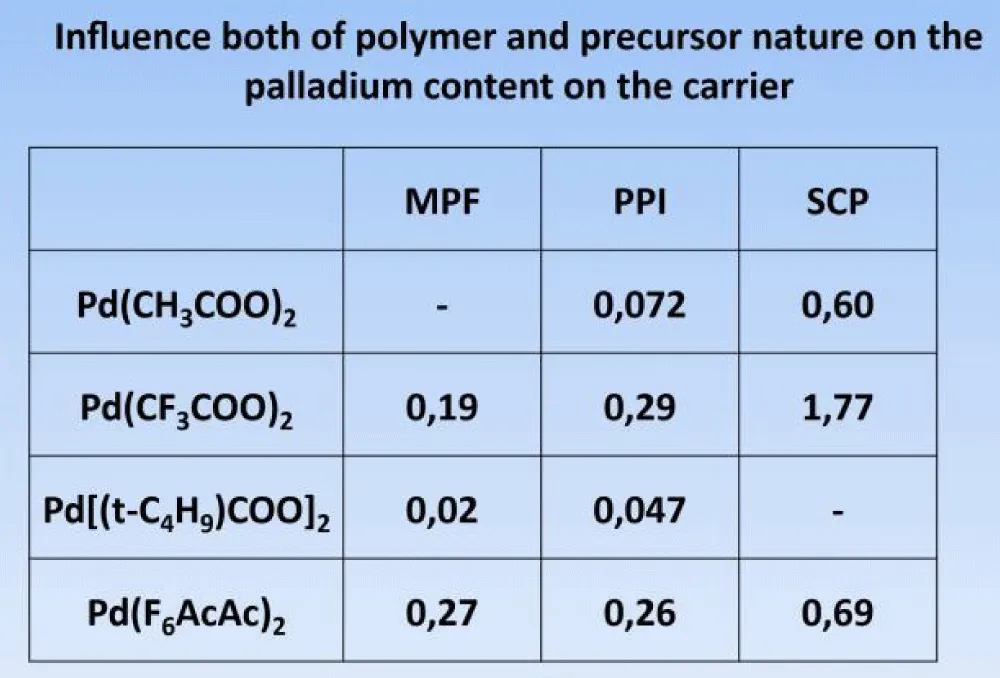
As previously shown (Figure 6), the amount of palladium strongly depends on the nature of the palladium complex, as well as on the polymer carrier, which used a phenol-formaldehyde polymer (MPF), polypropylene imine, and cross-linked polystyrene. (MPF), polypropylene imine (PPI) and cross-linked polystyrene (SCP).
Figure 6: Dependence of the palladium content (in wt. %) on the initial compound.
As can be seen from the figure, the content of palladium strongly depends on the nature of its starting compound. The presence of fluorine atoms increases the content of palladium, and the nature of the polymer carrier also helps to increase its content.
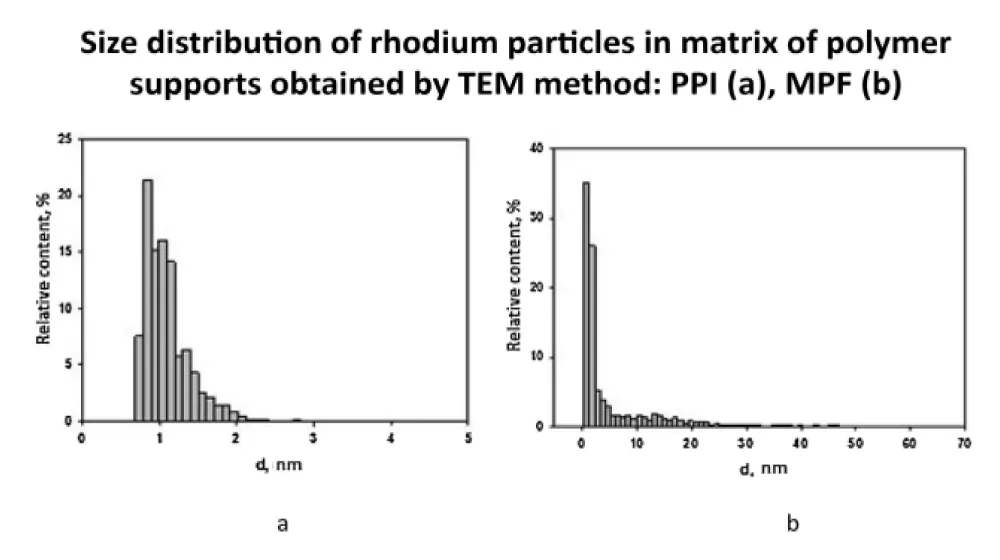
The following figure (Figure 7) shows the distribution of rhodium particles on two polymers, MPF and PPI (the initial compound Rh(Ac-Ac)3), determined by TEM [5]. The data obtained show that the sizes of rhodium particles are very small, for one of the polymers, PPI, they have 1.10±0.15 nm, for the second - 1.82 ± 0.14 nm, while there is practically a unimodal distribution in size.
Figure 7: Size of Rh particles in matrixes of polymer support PPI (a), MPF (b).
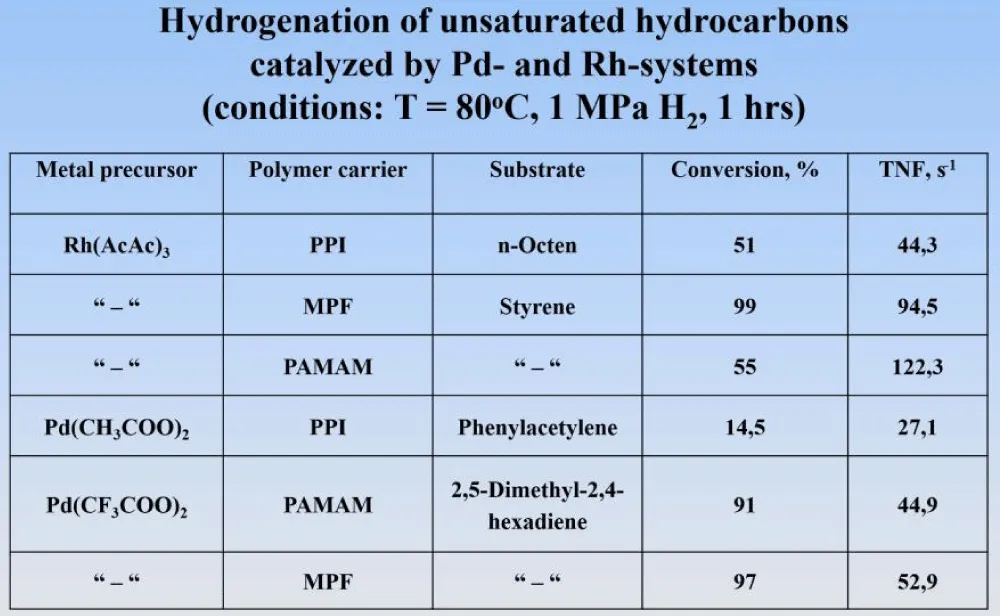
All the obtained catalytic systems were used for the hydrogenation of various organic substrates (olefins, conjugated dienes, and acetylenes) at a temperature of 80°C and a hydrogen pressure of 1 MPa for 1 hour (Figure 8). It can be seen that the conversion for rhodium is quite high, and the activity of the catalysts is expressed as the ratio of the moles of the reacted substrate to the number of metal g-atoms per unit time. For palladium catalysts, hydrogenation was carried out in the case of conjugated dienes and acetylenes, where hydrogenation occurs only when a single double bond in the substrate molecule is hydrogenated, resulting in the formation of olefins. The interest in this reaction stems from the need to purify monomer molecules (monoenes and their derivatives) from traces of dienes and alkynes, which act as catalytic poisons for their polymerization. The obtained data (GLC) allow us to conclude that palladium catalysts have increased activity and selectivity for olefin yield, which is about 97-99% of olefins with more than 94%conversion.
Figure 8: Hydrogenation of unsaturated hydrocarbons by Rd- and Pd-catalysts.
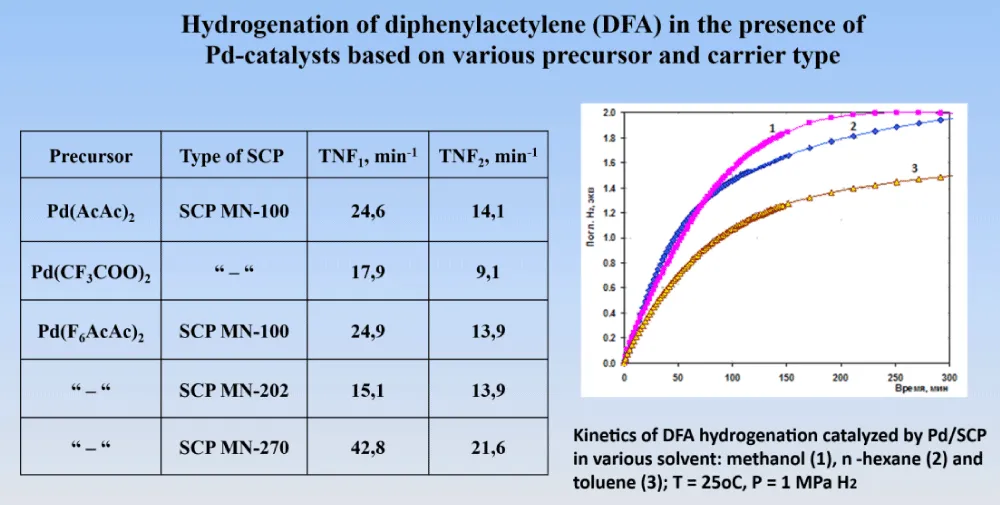
Further, during the hydrogenation of diphenylacetylene in the presence of a palladium catalyst, the deposition of palladium complexes on cross-linked polystyrene of various natures was studied (Figure 9) [6]. It turned out that when using the SCP MN-100 carrier, where the number of substituents includes tertiary amino groups, form more stable palladium catalysts. Hydrogenation was also tested in a number of different solvents, among which the most suitable was chosen toluene, in which the ratio of TNF1 to TNF2 was more preferable (3.75 in compared to 1.47).
Figure 9: Hydrogenation of diphenylacetylene in the presence of Pd-catalysts ba sed on various cross-linked polystyrene and carrier type.
We have developed another model, where the palladium atoms were obtained by laser focusing on a sheet of palladium, whose atoms immediately broke away from the plate and immediately landed on the nearby powder of aluminum oxide [7]. It should be noted right away that the whole process took place in an atmosphere of supercritical carbon dioxide. The source of radiation was a laser of the Lotis LS-2138TF brand (Belarus) with a wavelength of 1064 nm, a frequency of 50 Hz, a pulse duration of 15 ns and an energy of pulses of 160 mJ. The carrier was a powdered γ-aluminum oxide (fraction 0.25-0.50 mm). The temperature of the CO2 flow was 50 °C, and the pressure was 180 atm (Figure 10).
Figure 10: The scheme of the laser ablation of palladium atoms.
In the following figure (Figure 11), we can see that the traces of palladium atoms are just beginning to detach from the plate (a), then they move to the solid carrier (b), and finally they land on the carrier (c).
Figure 11: Photos of a palladium target in the process of laser ablation in SC-CO2.
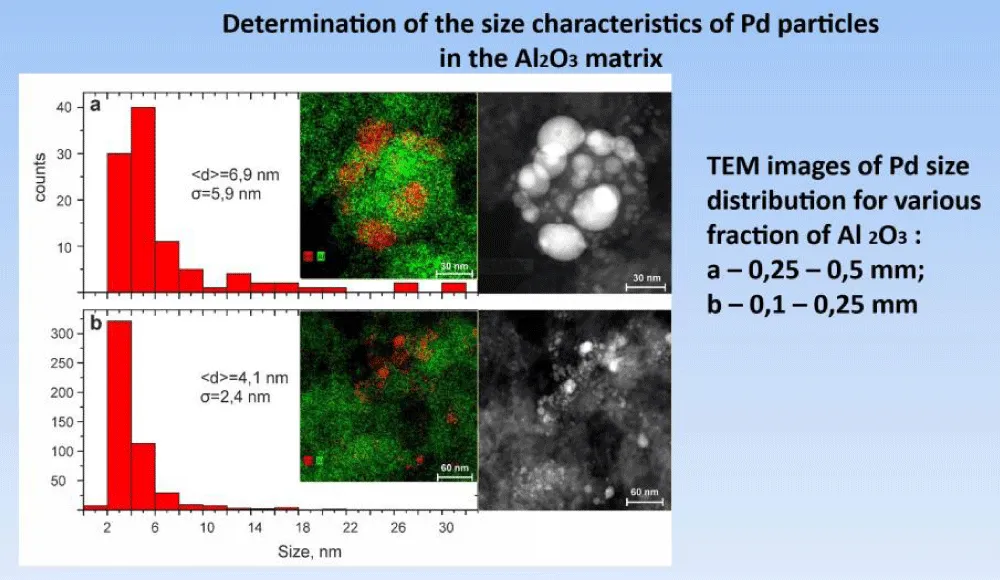
Next, the distribution of palladium particles on the surface of aluminum oxide was determined, and this was done for both the 0.25-0.5 mm and the 0.1-0.25 mm fractions of Al2O3 particles (Figure 12).
Figure 12: Determination of the size distribution for various fraction of Al2O3.
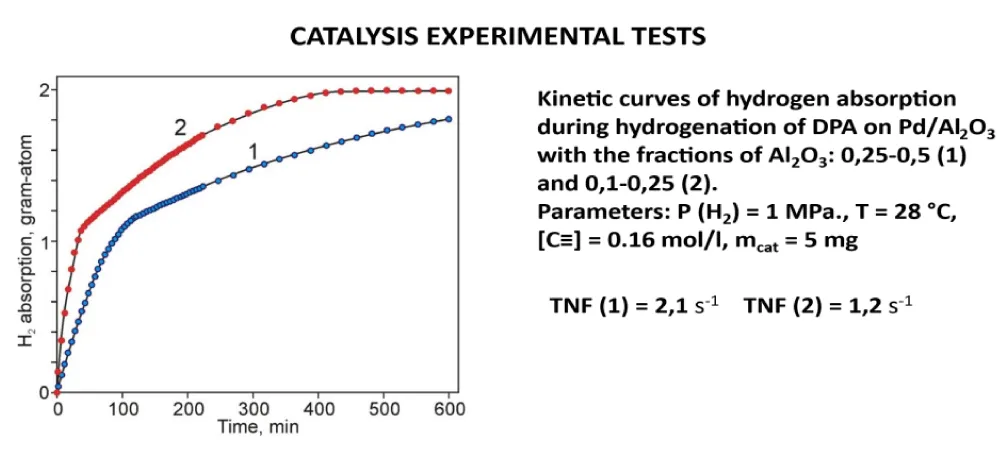
Then, the obtained catalysts were tested in the hydrogenation reaction of an acetylene derivative. Figure 13 shows the kinetics of the hydrogenation of this hydrocarbon, which indicates that the reaction proceeds very quickly, with a zero-order reaction in the first stage, suggesting a selective hydrogenation pathway for the acetylene compound (selective hydrogenation) [7,8].
Figure 13: Kinetic curves of hydrogenation of diphenylacetylene on Pd/Al2O3.
In summary, this study considered two fundamental methods for the synthesis of highly active nanocatalysts using supercritical media. The specific conditions and features of these synthetic procedures were detailed, highlighting how they provide effective immobilization of metals within the carrier matrix. Furthermore, various examples of heterogeneous catalysts were presented, alongside their dimensional characteristics and catalytic performance in reactions such as the hydrogenation of unsaturated compounds. Ultimately, the results obtained thus far demonstrate the significant prospects for developing new, highly efficient, and environmentally safe technologies for the production of heterogeneous catalysts across a range of petrochemical and organic synthesis processes.
- Zhang Y, Erkey C. Preparation of supported metallic nanoparticles using supercritical fluids: a review. J Supercrit Fluids. 2006;38:252–267. Available from: https://doi.org/10.1016/j.supflu.2006.03.021
- Satoshi Y, Yoko M, Takeshi F, Yoshihiro T, Katzuto O, Tomoya T, et al. Solubility measurements of noble metal acetylacetonates in supercritical carbon dioxide by high performance liquid chromatography (HPLC). J Supercrit Fluids. 2008;44(2):139-147. Available from: https://doi.org/10.1016/S0039-9140(96)02008-5
- Cross W, Akgerman A, Erkey C. Determination of Metal-Chelate Complex Solubilities in Supercritical Carbon Dioxide. Ind Eng Chem Res. 1996;35(5):1765-1770. Available from: https://pubs.acs.org/doi/abs/10.1021/ie950515c
- Smart NG, Carleson T, Kas T, Clifford AA, Burford MD, Wai CM. Solubility of chelating agents and metal-containing compounds in supercritical fluid carbon dioxide. Talanta. 1997;44:137. Available from: https://doi.org/10.1016/S0039-9140(96)02008-5
- Parenago OP, Timashev PS, Karakhanov EA, Maximov AL, Lazhko AE, Zolotukhina AV, et al. Obtaining of highly-active catalysts of unsaturated compounds hydrogenation by using supercritical carbon dioxide. J Supercrit Fluids. 2018;140:387-393. Available from: https://doi.org/10.1016/j.supflu.2018.07.010
- Lazhko AE, Bragina GO, Lyubimov SE, Davankov VA, Staheev AU, Parenago OP. Russ J Phys Chem B. 2021;14(4):1172.
- Parenago OP, Rybaltovsky AO, Epifanov EO, Bragina GO, Lazhko AE, Khmelenin D, et al. Synthesis of Supported Heterogeneous Catalysts by Laser Ablation of Metallic Palladium in Supercritical Carbon Dioxide Medium. Molecules. 2020;25(24):5807. Available from: https://doi.org/10.3390/molecules25245807
- Epifanov EO, Shubnyi AG, Minaev NV, Rybaltovsky AO, Yusupov VI, Parenago OP. Russ J Phys Chem B. 2020;14(3):64.