More Information
Submitted: January 23, 2026 | Accepted: February 07, 2026 | Published: February 09, 2026
Citation: Khan MI, Rahman F, Khurshid R, Mahfuza A, Siraj M, Rehman UU, et al. Biofunctionalisation of Nanoparticles: Emerging Strategies and Future Prospects for Biomedical Applications. Ann Adv Chem. 2026; 10(1): 008-032. Available from:
https://dx.doi.org/10.29328/journal.aac.1001061
DOI: 10.29328/journal.aac.1001061
Copyright license: © 2026 Khan MI, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestriCted use, distribution, and reproduCtion in any medium, provided the original work is properly cited.
Keywords: Nanomaterials; Surface modification; Medicine; Biomedical applications
Biofunctionalisation of Nanoparticles: Emerging Strategies and Future Prospects for Biomedical Applications
Muhammad Ijaz Khan1*#, Fazal Rahman2#, Razia Khurshid2,3, Anjum Mahfuza4, Muhammad Siraj5, Ubaid Ur Rehman2, Ihsan Ullah2, Ayaz Ahmad6 and Adnan Khan1
1Department of Biotechnology, University of Malakand, Chakdara, Pakistan
2Department of Biotechnology, Quaid-i-Azam University, Islamabad 45320, Pakistan
3Department of Biotechnology, Women University of Azad Jammu & Kashmir, Bagh, Pakistan
4Department of Environmental Science, Kunming University of Science and Technology, Kunming, China
5Institute of Biotechnology and Genetic Engineering, the University of Agriculture, Peshawar, Pakistan
6Center for System Biology, Tianjin Institute of Industrial Biotechnology, Chinese Academy of Sciences, Beijing, China
#These authors contributed equally to this work.
*Address for Correspondence: Muhammad Ijaz Khan, Department of Biotechnology, University of Malakand, Chakdara, Pakistan, Email: [email protected]
Nanotechnology has emerged as a transformative approach in modern medicine, offering powerful strategies for early disease detection and the development of safer, more precise, and cost-effective personalized therapies. Engineered nanoparticles, designed through controlled manipulation of size, shape, and surface chemistry, serve as highly efficient drug carriers that can improve therapeutic outcomes while minimising systemic toxicity. Surface biofunctionalization plays a pivotal role by enhancing nanoparticle stability, biocompatibility, and selective targeting. Functional ligands such as aptamers, peptides, and antibodies enable molecular recognition, facilitating accurate drug delivery and advanced diagnostic applications. Aptamers provide flexible, high-affinity targeting capabilities, whereas antibodies ensure specificity toward defined biomarkers. Despite existing challenges in clinical translation, biofunctionalized nanoparticles demonstrate significant promise in targeted drug delivery, diagnostic imaging, and theranostic systems. Antibody-conjugated nanoparticles have notably advanced diagnostics and treatment strategies for cancer, neurological disorders, and precision medicine. Similarly, transferrin-modified nanocarriers enable selective cellular uptake. Recent developments integrate therapeutic and imaging functionalities within unified nanoplatforms, allowing simultaneous drug delivery and real-time in vivo monitoring. This review highlights advances in surface engineering and biofunctionalization techniques, providing a comprehensive framework for designing multifunctional nanomaterials that synergistically combine diagnostic and therapeutic capabilities for next-generation biomedical applications.
Graphical abstract
Nanotechnology has received increased attention in the medical sciences due to certain health risks associated with conventional chemotherapeutics [1]. The existing successful chemotherapies have many limitations, for instance, adverse side effects, fast degradation in the body, high doses, low water solubility, and, most importantly, nonspecific targeting and drug resistance [2,3]. Conversely, small molecules are taken up by all types of cells in the body, preventing the medicine from delivering to the targeted site and increasing toxicity. In contrast, nanomaterials and nanoparticles may be conjugated with many chemical or biological materials, such as peptides, antibodies, or other small molecules, for target-specific drug delivery [2,4]. This nano-based approach can circumvent many drawbacks of chemical-based treatment modalities, including reduced toxicity, increased solubility, and release of specific targeted drugs at a particular temperature, pH, and light in the targeted region [5].
To overcome the difficulties posed by traditional medications’ pharmacokinetics, recent nanotechnology developments have concentrated on strengthening nanoparticle stability and boosting controlled drug release systems [6,7]. Theranostics is the method used for simultaneous treatment and diagnosis [8]. Nanobased-specific imaging probes have been developed to visualize, characterize, and quantify biological processes at the molecular level in a noninvasive way, leading to the accurate diagnosis of various diseases and individualised treatment [9,10]. Nanoparticles (NPs) functionalized with targeting ligands have also shown significant promise in the detection of early-stage cancers and in providing real-time insights into treatment efficacy [11]. These nanomaterials and nanoparticles have been functionalized (by modifying the surface or properties of the nanoparticles via the conjugation of specific functional groups) to improve their stability, biocompatibility, and targeting ability for targeted drug delivery and diagnosis [12,13].
Biofunctionalisation is an essential approach to functionalizing nanomaterials [2,14]. Functionalization approaches use chemical cross-linking reagents to conjugate imaging systems, natural phytochemicals, natural bioresources, bioinspired ligands, chemical agents, and therapeutic drugs [15]. In recent years, biofunctionalisation strategies have significantly improved using biomimetic peptides, antibodies, and nucleic acids for more specific targeting and enhanced bioactivity [16]. Different methods have been used for biofunctionalisation, including ligand-based biofunctionalisation strategies, stimuli-responsive biofunctionalization strategies, multifunctional biofunctionalization strategies (integration of therapeutic and diagnostic functions and dual-targeting approaches for enhanced specificity), and surface engineering and modification techniques. Moreover, incorporating innovative nanomaterials with adaptive properties in response to environmental stimuli has opened new avenues for personalised medicine [17].
This review provides a comprehensive and forward-looking analysis of emerging strategies for the biofunctionalisation of nanomaterials, with particular emphasis on advances achieved over the past decade. Unlike previous reviews that primarily summarise established functionalization techniques, this work critically integrates recent methodological innovations, compares their mechanistic advantages, and evaluates their translational relevance within clinically oriented biomedical contexts. We specifically highlight how modern surface engineering approaches address persistent limitations in targeted drug delivery, molecular diagnostics, and multifunctional theranostic platforms. In addition to surveying technological progress, this review uniquely examines the practical challenges governing clinical adoption, including reproducibility, scalability, biological performance and AI-assisted nanoparticle design. Particular attention is given to safety assessment, encompassing biocompatibility, immunogenicity, biodistribution, and long-term toxicity considerations associated with biofunctionalized nanoparticles. By incorporating safety and durability perspectives alongside engineering advances, this article provides a more balanced and application-driven framework than existing literature. Furthermore, we outline strategic directions for future research, emphasising robust fabrication methodologies, enhanced in vivo stability, and standardised evaluation protocols. Collectively, this review not only synthesises current knowledge but also establishes a structured roadmap for accelerating the safe and effective translation of biofunctionalized nanomaterials into next-generation biomedical applications.
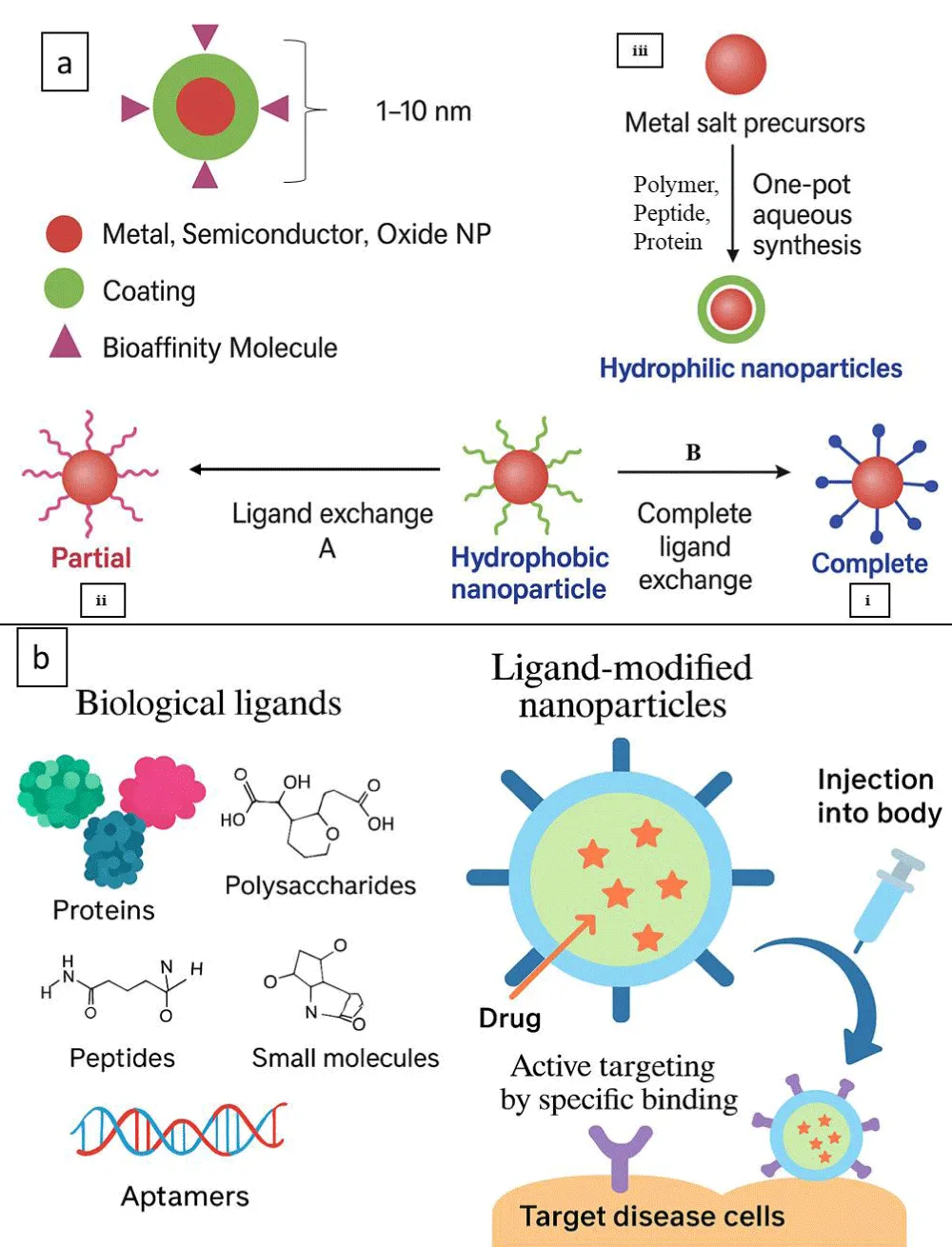
Nanotechnology advancements allow for the creation of nanoparticles with particular morphologies and the customisation of their surfaces to control their properties to suit different applications [18,19]. Therefore, there is considerable interest in the functionalization of NPs, their use in medicine, and their application in diagnosis. Over the past ten years, much research has been carried out on the synthesis and functionalization of NPs [20]. The NPs synthesis is divided into two categories based on phobicity, i.e., hydrophilic and hydrophobic. Hydrophilic and hydrophobic NP synthesis occurs when conditions in the aqueous phase are conducive to direct synthesis and hydrophobicity. The typical functionalization techniques consist of (i) Hydrophobic NPs are directly encapsulated in hydrophilic polymers, as shown in ligand exchange B, (ii) substitution of the primary surfactant ligand with thiols or other hydrophilic ligands, as shown in ligand exchange A, and (iii) interdigitated bilayer formation between polymers or amphiphilic molecules. The physical or chemical adsorption of appropriate ligands onto a surface is one of the most commonly used methods for engineering NP surfaces [21,22]. Often, coating techniques based on ligand exchange and chemisorption offer a superior option for customising the surface characteristics of NPs. NPs may exhibit extremely selective binding after fusion with proper targeted ligands, antibodies, or proteins, making them effective [23]. Many biological ligands have been identified and studied to facilitate the active targeting of NPs. Such biological ligands often bind to specific receptors on the target cells’ surface, increasing the cellular uptake of drug-containing NPs and therapeutic efficacy. Compared to singular ligands, an increased density of ligands is advantageous for promoting binding and cellular uptake through the multivalent effect [24]. Various types of ligands have been employed for this purpose, including proteins, polysaccharides, nucleic acids, peptides, and small molecules for biomedical applications, as shown in Figure 1.
Figure 1: a) Metal oxide functionalized nanoparticles for imaging applications. b) Illustration of biological ligands for active nanoparticle targeting for biomedical applications.
Specific binding of targeted ligands to disease biomarkers
One of the most promising approaches to better understanding disease biology and controlling disease is the use of disease biomarkers. A biomarker is a biological condition indicator. It can be an organic substance produced by aberrant cells, a protein, a protein fragment, DNA, or RNA. A disease biomarker is crucial for the early diagnosis and particular staging of a disease because it represents a “molecular signature” of the physiological state of a disease at a specific moment [25]. Chemical or biological forces can be detected using nanosensors, which partially function at the nanoscale. NPs conjugated to a directed ligand often form the basis of nanosensors. The ligand plays a crucial role in identifying the target marker, imparting specificity to the nanosensor. In contrast, the nanoparticle plays a dual role as both a signal generator and detector, ultimately determining the sensor’s sensitivity [26]. NPs have unique magnetic properties, high reactivity, improved electrical conductivity and strength, and a sizable surface area-to-volume ratio, all desirable and unmatched detection qualities. High-sensitivity nanosensors for biomarkers have been the subject of numerous investigations, which have ushered in a new era of early illness diagnosis and improved therapy [27,28]. An iron-binding glycoprotein called transferrin (Tf) transports iron throughout the body. Tf-modified NPs can target tissues and cells where Tf receptors are strongly expressed. One of the groups of researchers developed conjugated NPs to transfer therapeutic medications across the blood-brain barrier (BBB) by targeting Tf receptors on the blood side of the BBB [29].
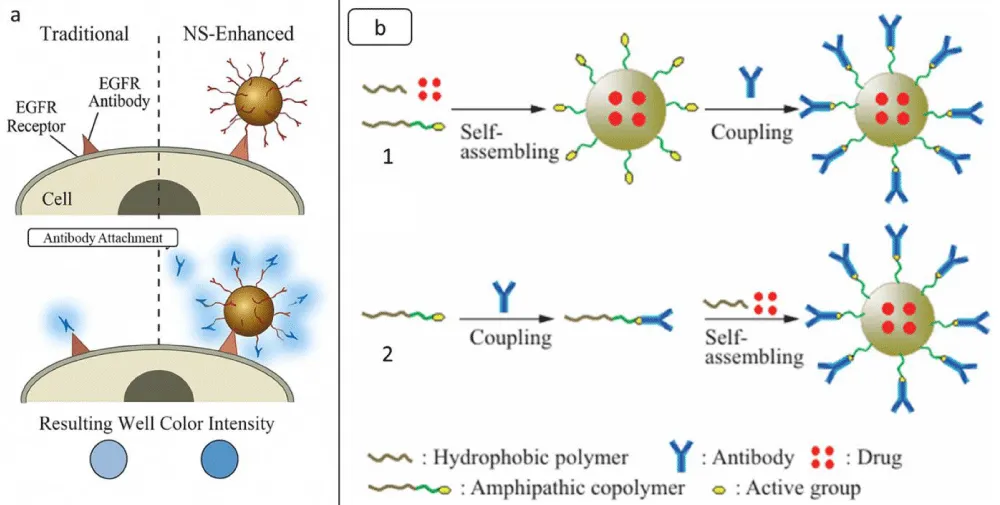
Antibodies and antibody fragments: In biology and medicine, antigen detection methods are crucial for diagnosing illnesses, assessing disease severity, and even predicting patient outcomes. The simplest method used worldwide for antigen recognition is ELISA [30]. In an ELISA, biological samples containing the target biomarker are treated with primary antibodies. Horseradish peroxidase (HRP)-conjugated secondary antibodies are bound by detectable primary antibodies [31]. Because antibody-coated NPs offer abundant binding regions for detectable secondary antibodies, studies have shown that the detection limit of ELISAs can be enhanced by using antibodies without NPs. They show this by employing either naturally occurring epidermal growth factor receptor (EGFR) or EGFR-Nanoshells antibodies in an ELISA with three cell lines with variable EGFR expression levels. Figure 2a [32] demonstrated that EGFR-NS, compared to unconjugated EGFR antibodies, might induce a 13-fold increase in signal intensity. The short size of antibody fragments decreases their immunogenicity and enables greater orientation of the targeted ligands and higher loading capacities [31]. Researchers have recently shown how anti-DEC205 ScFv fragments might target liposomes containing small interfering RNA (siRNA) in dendritic cells, leading to enhanced gene silencing in vivo. To reduce adverse effects on binding, their targeting ScFvs were coupled to the nanoparticles through a C-terminal cysteine created by site-directed mutagenesis. As a result, conjugation could take place far from the binding site. The authors highlighted the risks of employing full mAb-targeted nanoparticles in clinical settings. They mentioned that they could not use the full anti-DEC205 antibody for the same reason in unpublished preliminary results [33]. A long-standing objective of medicine is to safely encapsulate a mixture of harmful substances and administer them in a targeted manner. For this purpose, numerous intriguing candidates have already begun clinical trials, and nanoparticle-antibody conjugates have demonstrated considerable potential [34]. Formulating targeted NPs requires serial chemical processing. Generally speaking, the antibody could be conjugated onto the surface of the targeted NPs before or after formulation, which is called pre-conjugation and post-conjugation, respectively. Post-conjugation is the most commonly used method, which includes multiple steps, such as assembly of polymeric materials, activation of the NPs, antibody coupling, and purification. Briefly, the coupling and purification steps should be performed in a buffer solution to avoid the inactivation of these biomolecules in an organic solvent. However, this method often adds excess reactants to drive the conjugation reaction, thus making it difficult to get a uniform NP surface property and causing batch-to-batch variation [35], as shown in Figure 2b.
Figure 2: a. Schematic representation of standard and NS-enhanced ELISAs. After the primary antibodies have bound their target cell surface receptor, secondary antibodies are added in the usual ELISA (left), and the findings are then detected colourimetrically. The resulting signal is weak since few secondary antibodies are detectable for each receptor. In the NS-modified ELISA (right), NSs are coated with primary antibodies to provide many detectable secondary antibody binding sites, improving the colourimetric signal. b. Schematic illustration of the fabrication of antibody-conjugated NPs: post-conjugation (1) and pre-conjugation (2) adopted from [35] copyright@ Springer Nature 2020.
Peptides, peptide mimetic small molecules, and aptamers: A peptide mimetic is a substance that biologically resembles the active ligands of hormones, cytokines, enzyme substrates, viruses, or other biomolecules. The physiological activity of natural ligands may be antagonistically affected, stimulated, or otherwise modified. Several disease disorders that may be addressed with gene therapy have improved nonviral delivery of genes through peptide targeting. Targeted delivery genes have potential in therapies for cancer, CNS genetic disorder treatment, cystic fibrosis genetic correction, and other tissue-specific disorders, with cancer applications garnering the most interest [36]. A flexible method for producing functional materials for various purposes is the self-assembly of tailored synthetic peptides. Given the biological importance of their amino acid makeup, one plausible use for this material class is pharmacological and therapeutic delivery [37].
A few investigations successfully used peptide phage display to choose binders for small compounds such as fluorophores, microcystin-LR, and paclitaxel (Taxol). The phage display biopanning approach is employed to select specific peptides against nanomaterials. Nanotechnology can benefit significantly from these phage-displayed nanomaterial-binding peptides [38]. The targeting of the kidney and kidney cells, such as glomerular mesangial cells, glomerular endothelial cells, and tubule cells, has been performed using a range of targeting peptides and antibodies combined with nanoparticles. This approach may provide researchers with a wide range of potential targets when developing diagnostic or therapeutic approaches for renal disorders that impact certain kidney cell types [39]. Due to virus infection, altered dynein-binding peptides with NPs successfully facilitated transmembrane penetration across the BBB. This study emphasises the significance of the level of DBP alteration and its impact on facilitating targeted particle entry and offers a framework for further research on DBP-based nanocarriers [40].
Because of fewer composite structures and steric hindrance for biochemical assays, smaller molecules like amino acids and sulfhydryl compounds can be bioconjugated to the surface of AuNPs more easily and under control than biomacromolecules such as antibodies, enzymes, or polysaccharides [41]. Additionally, researchers have created several CuAAC-mediated immunoassays to recognise various illness indicators using functional AuNPs. Due to the availability of an extensive repertoire of antibodies that precisely bind proteins, nucleic acids, and small molecules, ELISA has been used extensively for detecting disease biomarkers, controlling food quality, and monitoring the environment [42]. An alluring substitute for cell surface protein detection is small-molecule-linked DNA. More than 50% of currently available medications target cell surface proteins. This substantial collection of small-molecule ligands is a possible toolkit for probing cell surface proteins. The advantages of DNA coupling include improved sensitivity and adaptability. It allows for sensitive detection and the programming of several processes, including recognition, catalysis, and encoding, to small-molecule ligands [43,44].
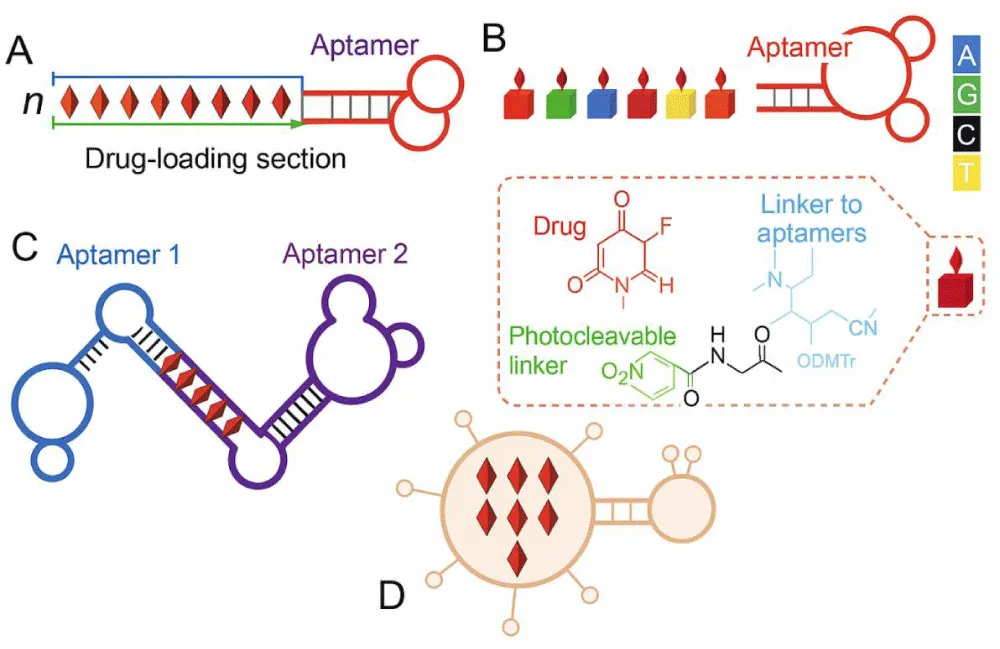
DNA or RNA single-stranded (ssRNA or ssDNA) aptamers have a distinctive tertiary composition that allows them to attach to their cognate molecular targets in a specific manner. A combinatorial chemical technique called “in vitro selection” or “in vitro evolution,” the Systematic Evolution of Ligands by Exponential Enrichment (SELEX), is used in molecular biology to produce oligonucleotides of single-stranded DNA or RNA that bind to a target ligand or ligands with specificity. This method is generally applied to screen aptamers with targets such as small compounds, biomacromolecules, diseased cells, progenitor cells, and cancerous cells [45]. Aptamers are continuously being explored as targeting ligands in the form of aptamer-drug conjugates (ApDCs) for the delivery of drugs. One of the standard cancer treatment options is chemotherapy. This has greatly enhanced cancer patients’ prognosis. The toxicity of conventional chemotherapy in healthy tissues is a common drawback. Thus, decreasing the time healthy tissues are exposed to these medications is anticipated to lower these adverse effects and increase therapeutic efficacy. ApDC-mediated targeted drug delivery, which delivers drugs to sick tissues or cells but not healthy tissues, has been investigated to accomplish this goal, as illustrated in Figure 3 [46]. Biomarkers can also be targeted with aptamers for diagnostic purposes. Researchers have created modified aptamer fluorescent silica NPs (FSNPs) to identify leukaemia cells by linking labelled amine Sgc8 aptamers to modified carboxyl FSNPs through amide conjugation [47]. The accurate and quick detection of infections, antibiotics, poisons, pollutants, and other substances or biomolecules has been achieved using aptamer-based biosensors [48]. To turn the biological reaction into a signal that can be detected, biosensors use aptamer specificity to target disease and the convertibility of the transducers.
Figure 3: ApDCs as chemotherapeutic agents. (A) An ApDC was designed to provide a manageable consignment of DNA-intercalating medications into targeted cancer cells by noncovalent complexation of aptamer-tethered nanotrains and pharmaceuticals. (B) A sample of programmable ApDCs was created utilising phosphoramidite containing a 5-FU prodrug through a photocleavable linker (inset: the molecular structure of phosphoramidite that incorporates a prodrug). (C) By quickly joining two different aptamers with a dsDNA linker, a bispecific ApDC, which is also used for drug delivery in bispecific drug loading, was created to mimic a bispecific antibody. (D) A diagram showing aptamer-functionalized nanocarriers for precise drug delivery.
Design considerations for ligand selection and conjugation
Numerous nanomaterials, including silica, polystyrene, gold, and biodegradable polymer nanoparticles, are biofunctionalizable. To make different bioconjugates, such as imaging agents, targeted systems of drug delivery, and diagnostic tools, it is essential to choose the right ligands and connect them. These processes are necessary for creating them [49]. These methods entail attaching a ligand (such as a peptide, aptamer, or antibody) to a carrier molecule (such as a nanoparticle, polymer, or liposome) to establish a particular interaction or functionality. The resulting bioconjugate’s success, stability, and efficacy depend on careful design considerations.
Affinity, specificity, stability, and biocompatibility: Affinity and specificity are key factors in molecular interactions. Affinity measures the strength of the bond between molecules, while specificity determines the molecule’s ability to bind to a specific target. In immunoprecipitation assays, affinity influences the effectiveness and cleanliness of the results, while specificity ensures binding to the desired target molecule. A ligand that specifically binds to the desired target was chosen. This reduces off-target effects and ensures that the conjugate primarily interacts with the intended chemical or cell type. The ligand should have a suitable affinity to target the desired entity effectively. A high affinity may make it difficult to separate from the target, whereas a low affinity may result in poor binding [50]. First, in developing immunoassays, the affinity and specificity related to antibody-antigen recognition have received much attention. To specifically and effectively bind different cancer cells, primary or secondary antibodies are covalently immobilised onto the NP surface.
The stability of the other parts of the conjugate, including the linker that connects the ligand and carrier molecules, must be considered. The resistance of the bioconjugate to changes in temperature and pH during handling, storage, and in vivo use was examined. The vulnerability of the bioconjugate to enzymatic degradation was analysed, paying special attention to whether the linker or carrier molecule is vulnerable to proteases or other enzymes found in the biological milieu [51]. The potentially harmful effects of the bioconjugate on cells should be examined, especially if it is utilised for in vitro research or as a treatment. It is unclear whether immunological reactions are caused by bioconjugates in living organisms. For prolonged applications or repeated dosages, this is especially crucial. The biocompatibility of any nanoparticle or other component with a changed surface can be examined if the bioconjugate is present [52].
By encapsulating W18O49 within PEGPCL NPs, scientists have effectively created W18O49@PEG-PCL NPs.
This encapsulation enhances the stability, biocompatibility, and internalisation of W18O49 NPs in cells. The resulting W18O49@PEG-PCL NPs exhibited excellent CT efficiency, low cytotoxicity, and remarkable photothermal effects. They can also be reserved for a long time, making them highly promising for future clinical trials. It is truly exciting to see their potential impact in the medical field [53]. The functionalization of the NP surface to enhance biocompatibility can be exploited by using different molecules; among these, PEG is one of the most used in vitro and in vivo applications [54,55], showed how the addition of PEG on the surface of Fe3O4 and SiO2 NPs increased the biocompatibility when PEGylated NPs were used at high concentrations (200 μg/mL) with murine neural stem cells, unlike bare Fe3O4 & SiO2 that caused a viability reduction of about 50 % already at a dose of 20 μg/mL. This study is a clear example of the highly convenient properties of PEG to increase NP biocompatibility. Furthermore, the presence of PEG on the NP’s surface also improves hemocompatibility, as observed with chromium-doped zinc gallate and diatomite-based NPs [56].
Conjugation methods and linker chemistry: Chemical conjugation, particularly bioconjugation, which involves forming stable covalent bonds between two molecules, at least one of which is a biomolecule, has evolved into a billion-dollar industry underpinning advancements in research, diagnostics, and therapeutics [57]. This versatile chemistry is the cornerstone for numerous biomedical applications of advanced functional nanomaterials. By combining the properties of multiple molecules within a single construct, conjugation strategies have enabled the development of therapeutics with enhanced pharmacokinetics and targeted delivery to specific cell populations, improved the biodegradability and efficacy of polymer-based drugs, heightened the sensitivity and specificity of diagnostic platforms, and facilitated the design of more biocompatible imaging agents and fluorescent probes [58]. Furthermore, precise chemical modification of biomolecules allows researchers to elucidate protein structures and functions, probe nucleic acid interactions, map receptor-ligand dynamics, and investigate the behaviour of carbohydrates and lipids. Building on these principles, innovative nanomaterials are continually being engineered, often using monomeric units such as nucleic acids or amino acids as fundamental building blocks, expanding the frontiers of nanobiotechnology and precision medicine [59].
The variety of compositions used to biofunctionalize NPs is enormous and considerably exceeds the scope of the current discussion [60]. Even with this heterogeneity, nearly all of these methods fall into one of three classes: (i) covalent chemistry, (ii) noncovalent interactions, and (iii) encapsulation [61]. The NMs themselves, along with their size, shape, and structure, the NM surface type, and the presence of functional end groups, in addition to the type of biomolecule, available functional groups, size, chemical composition, and what is eventually needed from the compounds in practice, determine the initial choice of which approach to use [62]. A wide array of functional groups such as amines, thiols (sulfhydryls) [63], alcohols (hydroxyls), carboxylic acids and their derivatives (including esters, anhydrides, and carbonates), along with aldehydes, ketones, alkenes, alkynes, haloalkanes, cyanates, isocyanates, isothiocyanates, azides, and others provides remarkable versatility for chemical conjugation [63,64]. By leveraging the distinct reactivity of these groups, researchers can rationally predict reaction pathways and design efficient synthetic strategies to prepare and conjugate a diverse range of molecules. For example, nucleophilic groups such as amines, thiols, and hydroxyls readily react with electrophilic species like carboxylic acids, esters, or activated olefins, forming stable linkages [65,66]. Likewise, unsaturated compounds, including alkenes and alkynes, can participate in addition and cycloaddition reactions, most notably exemplified by click chemistry, to yield robust and selective conjugates, as illustrated in Figure 4.
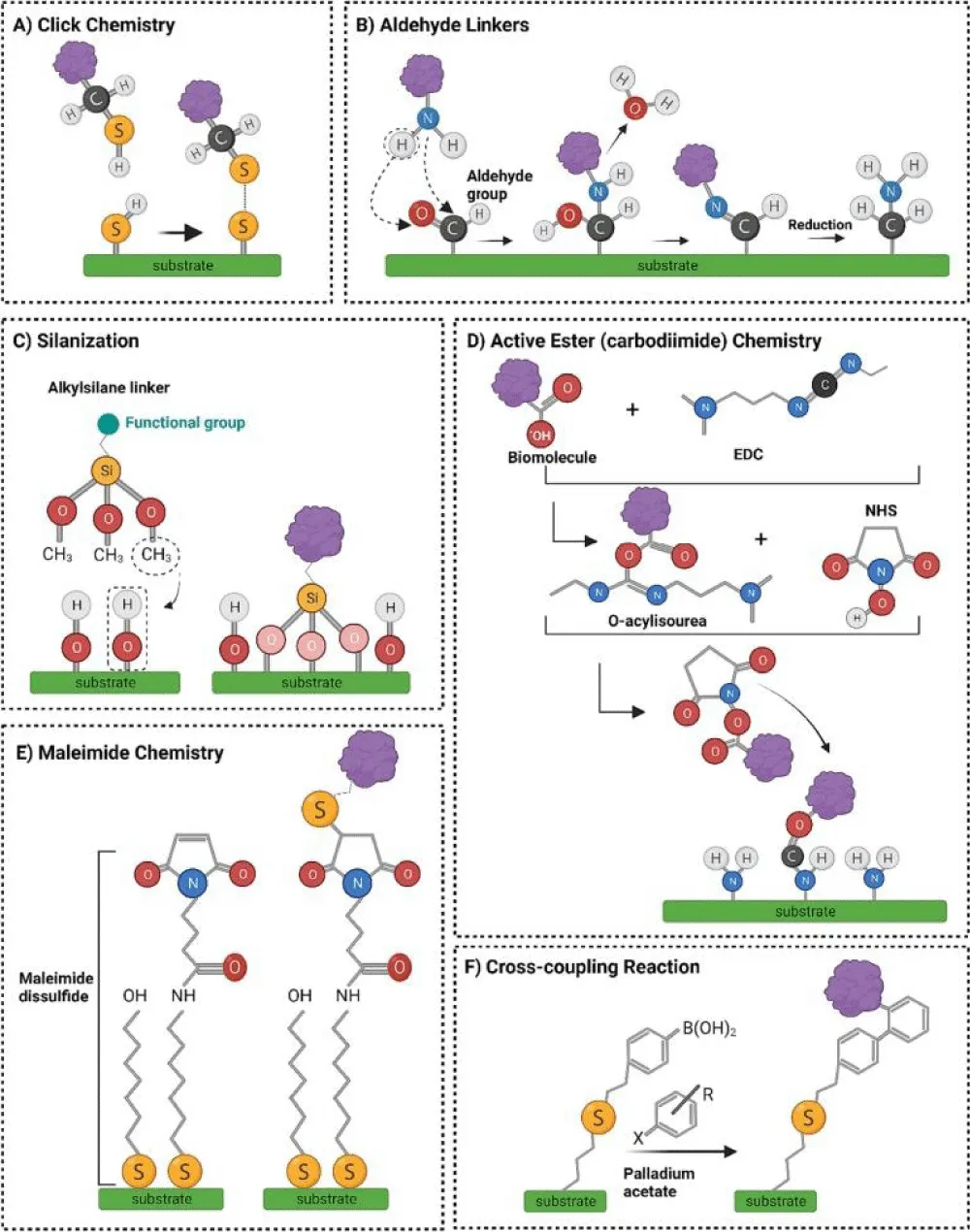
Figure 4: Schematic illustration of representative chemical reactions and popular surface bio-modification strategies discussed in this article. (A) Click reaction: thiol functional groups form transient disulfide (S–S) bonds, which can be cleaved under acidic conditions via reduction. (B) Covalent immobilisation of proteins onto substrate aldehyde groups involves the formation of reversible Schiff base linkages (imine bonds), which are then stabilised through reduction to form stable C–N bonds. (C) Silanization: Silane linkers undergo condensation reactions with hydroxylated surfaces, releasing methanol (CH₃OH) and creating robust siloxane bonds. (D) Carbodiimide-mediated active ester reaction: EDC (1-ethyl-3-(3-dimethylaminopropyl) carbodiimide) activates carboxyl groups to form an O-acylisourea intermediate, which reacts with NHS (N-hydroxysuccinimide) to generate an NHS ester, enhancing conjugation efficiency. The activated ester then reacts with amine groups on the substrate to form stable amide bonds. (E) Formation of self-assembled monolayers (SAMs) and subsequent bioconjugation using maleimide–thiol chemistry: maleimide-terminated alkane disulfides are assembled into SAMs, enabling selective binding to biomolecules containing free thiol groups. (F) Functionalization of mesoporous silica gels through organic coupling reactions: surface thiol groups react with 4-vinylphenylboronic acid via thiol-ene coupling, and Suzuki coupling with aromatic halides introduces additional functional moieties. Adopted from [67]. Copyright@2023 Elsevier
Most chemical-stimuli-responsive nanomaterials have been established based on disease conditions, such as inflammatory disorders, ischemia, tumours, and infections. Under these conditions, stimulus-responsive nanomaterials can benefit from particular micro-environmental alterations. Alternatively, they can react to externally provided physical stimuli, such as ultrasound, X-rays, magnetic fields, light, and temperature. Generally, exterior triggers are less variable and easier to manage than internal stimuli. The intended use, delivery site, therapeutic cost, and safety measures can influence the decision regarding which stimuli-responsive nanocarrier to use. Furthermore, numerous efforts have been made to improve the extensibility of different stimuli-responsive nanomaterials for enhanced treatment effects [68]. Using particular targeting agents, such as peptides, antibodies, small molecules, and nucleotide aptamers, to functionalize the surfaces of nanomaterials could dramatically enhance medication targeting. Another potential solution is the addition of groups or linkers that can respond to various extrinsic or intrinsic stimuli, which may make the NPs reactive to multiple stimuli and offer better platforms for highly advanced programmability.
pH-responsive nanoparticles
In recent years, pH and temperature fluctuations, in particular, have garnered much attention due to their physiological significance in the human body and practical advantages both in vitro and in vivo [69]. While ionizable functional groups (a weak acid or a weak base) cause the ionisation-deionisation of polymers in an aqueous solution to be out of balance, pH-sensitive polymers can react to changes in the surrounding medium’s pH by changing their structures and conformations. It is well known that the ideal pH for various bodily areas varies. A mildly acidic tumour microenvironment may be a target for pH-sensitive medication delivery. Due to their potential synergistic benefits, nanoparticles responsive to pH fluctuations under physiological conditions may be more appealing as cutting-edge drug carriers for cancer therapy. Recently, scientists have created hydrogels or beads made of pH-sensitive polymers for drug delivery. To our knowledge, research on pH-responsive polymeric nanoparticles is nevertheless quite rare. Nanoparticles provide polymers with unique features that distinguish them from bulk materials [70]. Introducing sustainable hollow cellulose NPs for regulating CaCO3 microspheres has led to exciting breakthroughs in biomineral materials. This innovative approach has successfully achieved sustained drug release, demonstrating the immense potential of combining hollow cellulose NPs with the theory of mineralisation. It is a brilliant prospect for the future. Moreover, porous CaCO3 microspheres have demonstrated outstanding potential as drug carriers. They exhibit high encapsulation efficiency, excellent pH responsiveness (with a pH of 2.0 significantly greater than 7.4), and good biocompatibility [71]. These properties enable precise control over drug release at specific locations. It is awe-inspiring.
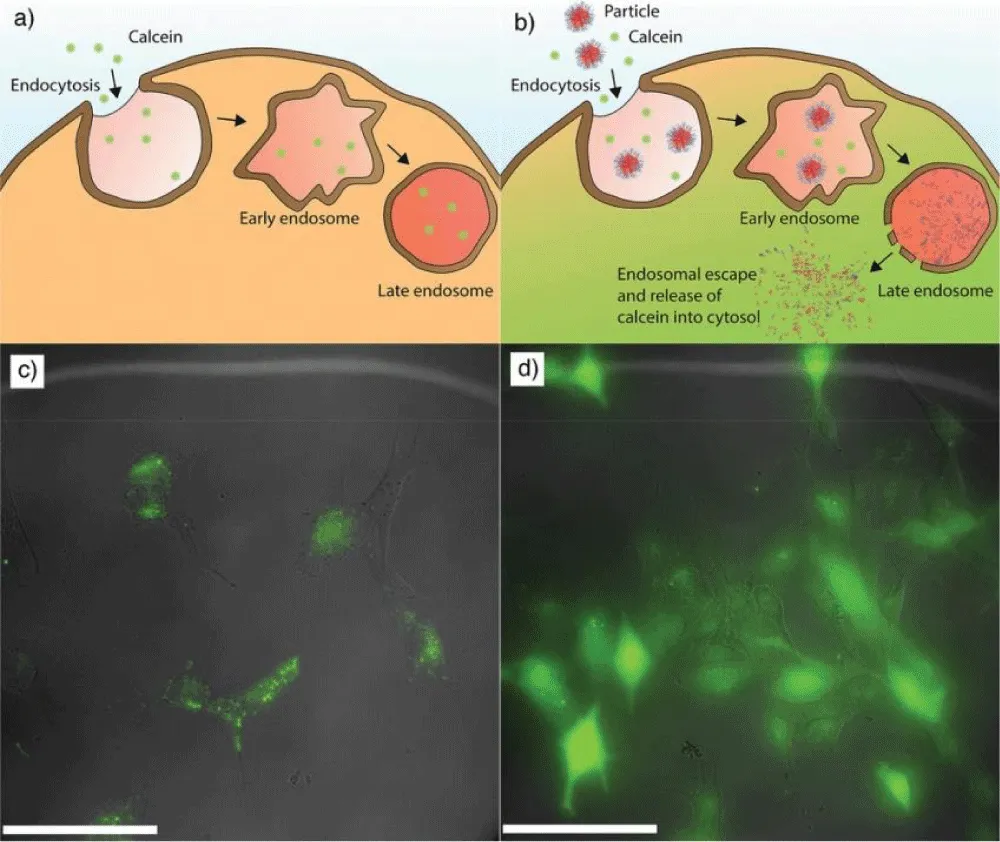
pH-sensitive polymers coating and drug release mechanism: Polymer nanoparticles provide a promising approach for improving drug delivery by protecting sensitive therapeutic agents until they reach their intended target. However, achieving adequate therapeutic delivery is complicated by several biological barriers. These difficulties include targeting specific cells and tissues, eluding the immune system, and delivering cargo to particular intracellular locations [72]. PH-responsive nanoparticles have gathered significant research interest due to the pH variations that occur during the endocytosis process within cells. The pH of the bloodstream is 7.4, the pH of the early endosomal compartment is 6.5, and the pH of the lysosomal compartment is less than 5 [73]. Nanoparticles can respond to pH changes by modifying their surface chemistry, shape, and size or disassembling and releasing their payload. Scientists have recently confirmed a one-step method for assembling nanoparticles using two pH-responsive polymers: the homopolymer poly (2-diethylamino) ethyl methacrylate (PDEAEMA) with a molecular weight of 38 kDa and the diblock copolymer poly (2-diethylamino) ethyl methacrylate-b-poly (ethylene glycol) (PDEAEMA-b-PEG) with a molecular weight of 16 kDa. The hypothesis suggests that PDEAEMA forms the hydrophobic core of the nanoparticles, while PDEAEMA-b-PEG provides stability under physiological conditions. These nanoparticles had an average size of 150 nm. The scientists explored the endosomal escape facilitated by the nanoparticles using calcein, a small fluorescent molecule. In the absence of nanoparticles, calcein (exhibiting green fluorescence) remains trapped within the endosomal and lysosomal compartments of the cell (Figure 5A-C). However, when pH-responsive nanoparticles help endosomes escape, calcein is released into the cytoplasm (Figure 5B-D). As a result, the calcein emits diffuse fluorescence inside the cell [70].
Figure 5: A) a diagram showing (green fluorescence) how calcein is concentrated in lysosomal and endosomal pouches without using NPs (carried for the control sample). B) a diagram illustrating how the addition of nanoparticles causes calcein to escape and release into the cytoplasm. C) Cellular microscopy images revealed that calcein was distributed punctually throughout the endosomal/lysosomal compartment, indicating that endosomes did not escape. D) Images from the cellular microscope reveal that approximately 30% of cells have diffuse calcein. Adopted from [70]. Copyright@2019, Wiley.
To improve the effectiveness of cancer treatment, another group of researchers employed charge-moving building blocks to create nanoparticles that are reactive to pH [74]. Scientists developed biosensors utilising a redox-activatable fluorescence sensor based on disulfide (qRAS). By conjugating the acceptor (Cy5) and insensitive pH fluorescence donor (TMR) onto the same cystine, one of these linkages, a disulfide bond, was used to create the sensor. Only one emission peak is observed since the donor fluorescence is muted in the off state. When the disulfide segment is broken down by glutathione (GSH), the donor’s luminance can be seen in the cell cytoplasm since the acceptor and donor are no longer nearby [75].
Temperature-responsive nanoparticles
A type of innovative material known as a thermoresponsive nanoparticle undergoes a phase transformation in response to a change in temperature. The critical solution temperature (CST) is the temperature at which a phase change occurs. CST knows a lower CST (LCST) if the thermoresponsive substance transforms from a hydrophilic and highly inflated condition to a hydrophobic and collapsed condition at CST after the temperature increases. CST is an upper CST (UCST) if thermoresponsive substances transform from hydrophobic and fall in condition to hydrophilic and highly inflated conditions at CST after an increase in temperature. LCSTs are typically thermoresponsive substances that have been studied for biomedical purposes. Thermoresponsive nanomaterials can be designed for various applications by adjusting their composition, LCST, architecture, and targeting segment [76]. Favipiravir was encapsulated in polymer-coated SNCs to facilitate its incorporation into well-defined films through layer-by-layer self-assembly, presenting a potential drug candidate for the treatment of COVID-19. Under the influence of a temperature stimulus, multilayer films made of polymer-coated SNCs and homopolymers of poly (methacrylic acid) (PMAA) displayed swelling and deswelling behaviours. The influence of steric hindrance on the assembly behaviour, swelling/deswelling transitions, and delivery efficiency of multilayer films composed of nanocapsules was investigated for the first time. The movie effectively contained favipiravir at 37 and 40°C, with as little as 50% released in 80 days; however, introducing a lower temperature of 25°C caused quicker favipiravir release [77].
Temperature-responsive nanoparticles
A type of innovative material known as a thermoresponsive nanoparticle undergoes a phase transformation in response to a change in temperature. The critical solution temperature (CST) is the temperature at which a phase change occurs. CST knows a lower CST (LCST) if the thermoresponsive substance transforms from a hydrophilic and highly inflated condition to a hydrophobic and collapsed condition at CST after the temperature increases. CST is an upper CST (UCST) if thermoresponsive substances transform from hydrophobic and fall in temperature to hydrophilic and highly inflated conditions at CST after an increase. LCSTs are typically thermoresponsive substances that have been studied for biomedical purposes. Thermoresponsive nanomaterials can be designed for various applications by adjusting their composition, LCST, architecture, and targeting segment [76]. Favipiravir was encapsulated in polymer-coated SNCs to facilitate its incorporation into well-defined films through layer-by-layer self-assembly, presenting a potential drug candidate for the treatment of COVID-19. Under the influence of a temperature stimulus, multilayer films made of polymer-coated SNCs and homopolymers of poly (methacrylic acid) (PMAA) displayed swelling and deswelling behaviours. The influence of steric hindrance on the assembly behaviour, swelling/deswelling transitions, and delivery efficiency of multilayer films composed of nanocapsules was investigated for the first time. The films effectively contained favipiravir at 37 and 40°C, with as little as 50% released in 80 days; however, introducing a lower temperature of 25°C caused quicker favipiravir release [77].
Thermosensitive polymers, hydrogels, and drug release strategies: The creation of thermoresponsive nanomaterials has made use of a variety of thermoresponsive polymers. Thermoresponsive polymers, such as poly (N-substituted acrylamides) like poly (N, N-dimethyl acrylamide) and poly (N-isopropyl acrylamide) (PNIPAAM), represent a distinct class of materials. PNIPAAM, the first and most extensively studied thermoresponsive polymer, exhibits a lower critical solution temperature (LCST) of 32 °C, close to the physiological temperature of 37 °C. The molecular weight, concentration, or other environmental factors do not affect the LCST of PNIPAAM [78]. Poly(N-vinyl-alkylamine)s, such as poly(N-vinylisobutyramide) and poly(N-vinyl caprolactam), represent a second class of thermoresponsive polymers with LCSTs ranging from 30 °C to 50 °C. Intestinal Caco-2 cells from humans and bronchial Calu-3 cells tolerated poly (N-vinyl caprolactam) well, but less research has been done on using poly (N-vinyl caprolactam) than PNIPAAM. Pluronics block copolymers of poly (propylene oxide) and poly (ethylene oxide) constitute the third class of thermoresponsive polymers. The lengths and ratios of hydrophobic poly (propylene oxide) and hydrophilic poly (ethylene oxide) segments can be adjusted to produce LCSTs between 20 and 85°C. They are amphiphilic polymers that have been given FDA approval for use as medicinal and food additives. In addition to the synthetic polymers mentioned above, thermoresponsive polymers can contain polypeptides or lipids [79].
Thermoresponsive nanomaterials, known as the LCST, are of exceptional quality and can be used to localise medications at a specific spot. Different thermoresponsive nanomaterials have LCSTs that are lower or greater than body temperature (37 °C), and they are constructed of various thermoresponsive polymers with various chemical properties. Thermoresponsive nanocarriers with LCSTs below 37 °C undergo a sol-to-gel phase transition at physiological temperature, enabling extended drug release and enhanced permeability across biological barriers. For instance, self-assembled poloxamer 407 thermoresponsive nanogels increase muscone permeability across the cornea by 3.4-fold upon attachment to the ocular surface [80]. Following injection into Sprague‒Dawley (SD) rats, the hydrophobic poly(propylene oxide) and hydrophilic poly(ethylene oxide) components of Pluronics1, along with self-assembled succinate d-α-tocopheryl PEG, formed micelles or vesicles at 50°C, facilitating the passage of the small molecular model drug Rho123 through the blood-brain barrier and enhancing its permeability [81]. Nanoparticles with an LCST slightly above 37°C remain soluble in physiological fluids and can circulate within the body at normal body temperature. However, thermoresponsive nanomaterials in the bloodstream become hydrophobic and penetrate surrounding tissues and cells more effectively when the target site is internally heated to 40 °C - 42 °C using methods such as ultrasound, magnetic fields, radiofrequency, or near-infrared (NIR) light [82]. This approach facilitates thermally targeted drug delivery, as demonstrated by the effective encapsulation of drugs such as doxorubicin, 5-fluorouracil, and 17-(allylamino)-17-demethoxygeldanamycin into various carriers, including cationic thermosensitive liposomes, core-shell thermoresponsive nanoparticles, and chitosan-g-poly(N-vinyl caprolactam) thermoresponsive nanoparticles [83].
Enzyme-responsive nanoparticles
Designing nanoparticles whose physical characteristics respond to an enzyme’s biocatalytic action is a developing field in bioresponsive nanomaterials [84]. In drug research and therapy, enzymes are crucial targets because they are essential for cell control. The nanomaterial can be configured to transport pharmaceuticals through the enzymatic transition of the transporter when the enzymatic function is linked to a specific enzyme or tissue that is present in larger amounts at the target area. Additionally, as deregulation of enzyme expression is a hallmark of many disorders, observing enzyme function can be a helpful diagnostic tool [85]. Using reliable immobilisation techniques for changing nanoparticle surfaces and the creation of ligands that convert the enzymatic function into a structural change in the NP solution are essential steps in manufacturing these nanoparticles [86]. The overexpression of matrix metalloproteinases in specific cancer types enhances the bioavailability of chemical drugs and enables precise therapeutic targeting. To improve biological selectivity and reduce side effects, researchers have developed smart drug carriers composed of enzyme-responsive nanoparticles using a functionalized copolymer (mPEG-Peptide-PCL) sensitive to matrix metalloproteinases [87].
Enzyme-cleavable linkers, prodrugs, and drug release mechanisms: Modified enzyme-activatable anticancer prodrugs consist of an antitumor agent, a divisible linker, and a functional component. By masking the cleavable linker, a prodrug composition can produce many functionalities that conventional medications cannot carry out and decrease the lethality of typical antitumor medications. Once the prodrug is metabolised, its cytotoxicity is selectively restored through specific chemical reactions within the cancer microenvironment [88]. In lysosomes, beta-galactosidase (β-gal) hydrolyses galactosides into monosaccharides and catalyses the breakdown of glycosidic linkages. Numerous gal-activatable prodrugs have been created due to the reported overexpression of gal in malignancies [89]. Burkholderia pseudomallei, the cause of melioidosis, and other CDC-classified Tier 1 disease-causing agents that cause pulmonary intracellular infections necessitate prompt medical treatment with localised and sustained endogenous transport of concentrated medicines. By enzymatic drug release and the integration of mannose-targeting ligands, researchers have shown that a polymeric antibiotic prodrug platform may be developed to improve the release of endogenous PK drugs [90].
The assembly and disassembly of nucleic acid ligands in the presence of enzymes are described for a nucleic acid-functionalized nanocapsule. Because of an incorporated ester cross-linker in the particle’s core, the particles are entirely degradable in reaction to esterases. Nanocapsules can encapsulate hydrophobic small molecules during synthesis and, following self-assembly, can be stabilised through covalent cross-linking using copper-catalysed click chemistry [91]. In vivo experiments using a mouse model of hepatocellular tumours revealed that amphiphilic drug-loaded biomimetic phospholipid-like NPs had greater anticancer and tumour-targeting effects. The targeted release of the nanoparticle 1-O-octadecyl-2-conjugated linoleoyl-sn-glycero-3-phosphatidyl gemcitabine was achieved through the action of sPLA2, PLC, and PLD during tumour tissue division [92]. Scientists have developed innovative up-conversion nanoparticle-loaded phosphate micelles designed for targeted prostate cancer therapy, which can be cleaved by the sPLA2 enzyme [93]. Contact lenses based on hydrogels loaded with nanoparticles can be employed for ocular medication transport. Chitosan-poly(acrylic acid) nanoparticles, in situ gelled nanoparticles, and cellulose nanocrystals (CNCs) in PVA lenses were used to create two possible contact lens surfaces for regulated eye medication transport [94].
As researchers look into the intricate relationships between biology and engineering, multifunctional biofunctionalisation technologies are at the forefront of innovation. These methods provide fascinating opportunities for addressing challenges in the medical and other industries. A potential advancement in biomedicine is incorporating therapeutic and diagnostic functions into multifunctional biofunctionalisation techniques. The coexistence of therapeutic and diagnostic capabilities is made possible by these techniques, which offer several advantages for personalised medicine. Due to these techniques, the early detection of diseases and the accurate delivery of targeted therapies are now achievable. These techniques improve patient outcomes while lowering the cost of other procedures and treatments, thereby increasing the standard of healthcare. Moreover, details on the integration of therapeutic and diagnostic functions are provided below:
Combination of drug delivery and imaging agents
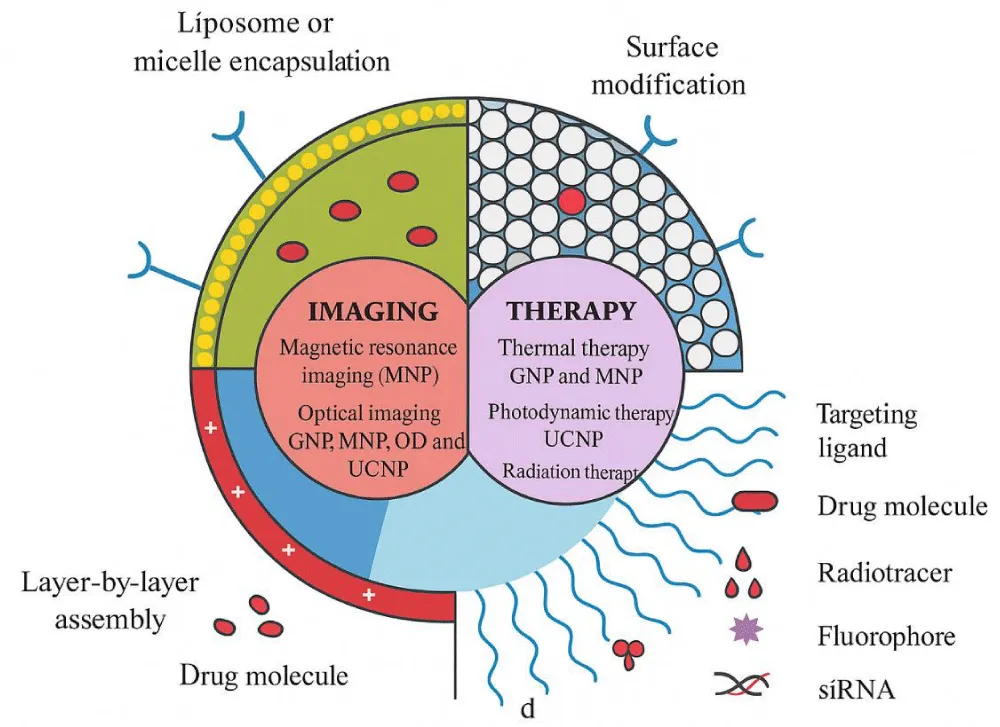
NPs are colloidal particles that deliver drugs to precise locations [95]. Nanoparticles transport biomolecules, including enzymes, peptides, genes, proteins, vaccines, and small-molecule drugs, via multiple delivery routes such as pulmonary, vaginal, intranasal, buccal, and oral pathways. They are widely used in treating cancer, neurological disorders, gene therapy, metabolic disorders, immunotherapy, tissue regeneration, and theranostics [96]. Advancements in nanotechnology and the growing demand for biomedical applications have driven the development of multifunctional nanoparticles. Through nanocrystalline synthesis, advanced polymer techniques, and surface functionalization, these nanoparticles can combine multiple capabilities, including thermal therapies, imaging contrast enhancement, and targeted gene or drug delivery. While still in its early stages, the field of multifunctional nanoparticles holds significant potential in medical advancements such as image-guided therapies, multimodal imaging, and theranostics [97]. More importantly, nanocrystals can be readily decorated with several generic coating strategies, including silica/mesoporous silica, micelle, liposome, and layer-by-layer assembly of biopolymers. These coating methods are helpful for both the physical/chemical adsorption of small molecules at substantial payload ratios and the encapsulation of multiple nanocrystals [98]. As such, numerous multifunctional nanoparticles can be generated by combining existing nanoparticles with small molecules, as shown in Figure 6.
Figure 6: Schematic diagram of multifunctional nanoparticles. Multifunctional nanoparticles can be generated by combining nanocrystals with different functionalities or combining nanocrystals with functional small-molecule cargos through different surface engineering strategies. Four typical coatings developed for inorganic nanocrystals are (a) liposome or micelle encapsulation, (b) mesoporous silica coating, (c) layer-by-layer assembly, and (d) surface conjugation. Abbreviations: GNP, gold nanoparticles; HfO, hafnium oxide nanoparticles; MNP, magnetic nanoparticles; QD, quantum dot; UCNP, upconversion nanoparticles.
Various techniques are being developed to synthesise AuNPs with functional moieties, improving their interaction with biological molecules and enhancing their effectiveness as drug carriers with greater specificity. Recent methods for AuNP functionalization include the use of either one or a grouping of functional groups such as antibodies, amino acids and polypeptides, receptors, oligonucleotides, bovine serum albumin (BSA), oligo or polyethene glycol (PEG), and various similar particles [99]. These carriers can carry drugs by adsorbing, entrapping, or covalently bonding to them. An optimal nanoparticle-based drug delivery system should efficiently locate, identify, bind to, and deliver therapeutic agents to diseased tissues while minimising or avoiding harm to healthy tissues [100]. Thus, encapsulating the targeted ligand(s) on the nanoparticle surface is the most popular method. These targeting ligands may include peptides, nucleic acid aptamers, antibodies, small molecules, and designed proteins. The drug is dissolved, encapsulated, entrapped, or bound to a nanoparticle. Recent advancements have focused on coating synthetic polymeric nanoparticles with cellular membranes for biofunctionalisation. This cell membrane-camouflaged nanoparticle platform offers a novel delivery strategy with the potential to enhance therapeutic efficacy in treating various diseases [101,102]. Peptides can functionalize gold nanoparticles through chemical conjugation, ligand exchange, and chemical reduction. The aggregation of colloidal gold nanoparticles can be stabilised or destabilised using specific peptide sequences responsive to environmental factors such as pH or temperature. These systems have broad potential applications in targeted drug delivery platforms and colourimetric sensing, including biosensing [100].
Hybrid hyaluronic acid and dextran structures were used to coat magnetic nanoparticles, demonstrating excellent biocompatibility. The biological results of cisplatin functionalization underwent a significant change, making these particles powerful drug delivery vehicles with the required potency against cancer cells [103]. Recent studies have demonstrated that fucoidan-functionalized nanoparticles hold potential as targeted agents for drug delivery and molecular imaging applications [104]. Magnetic nanoparticles (MNPs) have emerged as versatile theranostic tools in biomedical applications, including innovative therapeutics, diagnostic imaging, and drug delivery. Both clinical and preclinical studies have extensively investigated their functionalization, controlled drug release, targeted delivery, and image-guided capabilities [105]. Metal nanoparticles have also been studied for their use in CT imaging. Au nanoparticles can be used for imaging due to their ability to attenuate X-rays [106,107].
The future of biomedicine has much to gain from combining imaging agents and nanoparticle drug delivery. These adaptable nanoparticles provide several advantages, including penetrating biological barriers, reducing side effects, and boosting therapeutic effectiveness. Carefully focusing on specific tissues or cells minimises damage to healthy surrounding tissues, making treatments more effective and minimally invasive. Healthcare workers can monitor the progress of new medications, assess patient responses, and make necessary adjustments thanks to real-time imaging capabilities, which leads to more customised and adaptable treatment plans [108]. As this field of study matures, there is endless potential for improvements in patient care, therapies, and diagnostics, leading to a better and more personalised future for healthcare.
Imaging modalities
Imaging techniques encompass optical microscopy, photoacoustic imaging, ultrasound, magnetic resonance imaging (MRI), positron emission tomography (PET), X-ray computed tomography (CT), and single-photon emission computed tomography (SPECT) [109]. Nanoparticles play a vital role in multifunctional molecular imaging and multimodal applications. Materials such as manganese oxide (MnO), superparamagnetic iron oxide (SPIO), quantum dots (QDs), and gold nanoparticles or nanorods exhibit unique properties, including superparamagnetism, photoluminescence, paramagnetism, and surface plasmon resonance (SPR). Multifunctional nanoparticles (MNPs) can be engineered for targeted imaging by modifying their surface or attaching specific affinity ligands. These MNPs are utilised across various imaging modalities, such as MRI, CT, SPECT, photoacoustic imaging (PAI), PET, ultrasound, and fluorescent or two-photon imaging [110,111].
Functionalized fluorescent Np-based imaging methods possess the potential to address such objectives via noninvasive and accurate time visualisation of biological activities in vivo. For instance, Xie et al. developed a specialised lipid nanobubble for T lymphocyte detection using ultrasound (US) imaging. These targeted nanobubbles have been shown to provide a noninvasive method for detecting acute rejection [112]. Fluorescent agents based on nanoparticles have been developed to target many of the same biological markers as small molecules and macromolecules. Their distinctive material properties and nanoscale dimensions make them suitable for specific molecular imaging applications [113]. The highest spatial resolution for diagnosing microscopic diseases is possible using fluorescence imaging technology, particularly near-infrared (NIRF) imaging. Its clinical utility is still limited by its limited penetration depth, autofluorescence, and scattering characteristics in different tissues. Potential blink and photobleaching effects, limited fluorescence in the target lesion, and other factors may lead to low sensitivity for detecting abnormalities. Furthermore, a more considerable uptake in target lesions is made possible by the extended stay in circulation [114].
Three-dimensional anatomical images produced by X-ray computed tomography (CT) are valuable for diagnosing ischemia, tumours, brain injury, and other diseases. This approach is based on changes in X-ray beam absorption in different tissues. It is relatively cost-effective and widely available in hospitals. The lack of functional data and low soft tissue sensitivity are significant drawbacks of CT. The soft tissue sensitivity can be compensated for by combining imaging with MRI. More frequently, CT contrast agents that significantly absorb X-rays are utilised to improve selectivity, sensitivity, and the ability to distinguish between normal and diseased tissue. These included iodinated agents, TaOx, and gold nanoparticles [115].
Dual-targeting approaches for enhanced specificity
Dual targeting is a method that increases the concentration of NPs within the TME (tumour microenvironment) and enhances the effectiveness of targeting cancer cells. This approach combines purely biological techniques with physicochemically responsive innovative delivery strategies. In both methods, either a single ligand is utilised to target a single receptor on various cells, or two different ligands target multiple receptors on the same or other types of cells [116]. Researchers have described the creation of an immunosuppressive tumour microenvironment-beating NP platform. These NPs are coated with distinct antibodies that work in concert with both T cells through the 4-1BB costimulatory pathway and block inhibitory PD-L1 signalling. Immune switch nanoparticles significantly slow tumour growth and enhance survival in various in vivo colon cancer and murine melanoma models, outperforming soluble or nanoparticles independently coupled with inhibitory and stimulatory antibodies. These particles bypass the requirement for prior knowledge of tumour antigens and enhance the conjugation between effector and target cells. Their use has improved the specificity, density, and functionality of tumour-infiltrating CD8+ T cells in vivo [117].
Multiple ligands for the simultaneous targeting of different receptors: NP-based Drug delivery systems (DDSs) have been widely explored for treating several diseases, particularly cancer. An additional layer of sophistication is often incorporated in NP design via an active targeting technique to increase the effectiveness of antitumor drugs [118]. Since cancer cells typically overexpress many receptors on their surface, a dual-ligand targeting strategy can improve the targeting ability and increase nanoparticle delivery into cancer cells. Researchers have thoroughly examined the cellular uptake of dual-ligand-coated nanoparticles using DPD (dissipative particle dynamics) simulations. NPs adorned with long dual ligands recognise some rearrangements during engulfment, which may be advantageous for the complete engulfment of nanoparticles [119]. Through ligand functionalization, nanoscale delivery systems can control or trigger release and passive and active targeting. A practical approach for targeting cancer cells involves functionalizing nanocarriers with ligands that bind to overexpressed receptors, such as the transferrin receptor and epidermal growth factor receptor (EGFR). These receptors often serve as cancer biomarkers, and extensive research has identified ligands capable of enabling active targeting. Among the earliest molecules used for nanoparticle targeting was folic acid, with recent advancements enhancing its effectiveness as a cancer-targeting ligand. For example, Yang et al. developed lipid-coated calcium phosphate nanoparticles (LCPs) loaded with a pool of siRNAs. These nanoparticles were further coated with the tumour-targeting ligand anisamide and PEGylated lipids, a strategy widely applied in cancer-targeted nanomedicines [120].
Enhanced targeting efficiency and therapeutic efficacy: One of the key strategies for reducing side effects and increasing therapeutic efficacy is targeted drug delivery of therapeutic agents. Enhancing cellular internalisation and tissue retention/accumulation should be considered when designing NPs for targeted drug delivery [121]. Currently, nanoparticles may successfully deliver nucleotides, recombinant proteins, and vaccines. Gatti et al. produced NPs based on chitosan/dextran sulfate formed through polyelectrolyte condensation for insulin administration. The encapsulation prevented the partial breakdown of insulin and demonstrated sustained release, indicating effective mucus complexation between the NPs and mucin [122].
Polymer-coated NPs have been employed to accelerate biodegradation kinetics. PEG (polyethene glycol) was utilised to coat the surface of the NPs. The resulting NPs increased blood drug concentrations in the brain, intestine, and kidney by escaping the reticuloendothelial clearance system [123]. Enhancing the absorption and accumulation of nanoparticles in target cells is crucial for achieving greater treatment efficacy. This can be accomplished by surface functionalization with specific targeting ligands or through co-administration with other types of nanoparticles.
Additionally, several bioinspired molecules considerably impact the cellular uptake of NPs. Russell-Jones, et al. demonstrated that the conjugation of NPs with vitamin B12 vastly increased their uptake into an intestinal cell line, Caco-2 cells, compared to that of uncoated NPs. Yin Win, et al. utilised vitamin E to coat the surface of PLGA nanoparticles, significantly enhancing their adhesion ability and cellular uptake into Caco-2 intestinal cells [124]. Drug-loaded NP carriers can avoid the rapid breakdown of drugs via the body’s digestive enzymes. Drug-loaded NP carriers can change the transport membrane, increase the permeability of drugs in biofilms, and enhance drug uptake in the cell. The original drug can be modified using NPs as carriers to increase its water solubility or acquire a targeted and sustained release function, thus increasing its anticancer effectiveness and decreasing its side effects [125]. Mesoporous SeNPs can deliver doxorubicin to breast cancer patients with less toxicity and potent anticancer effects. Functionalized SeNPs with curcumin have been shown to have enhanced chemopreventive efficacy [126]. Surface modification of purified exosomes offers a promising approach to improve their targeting efficiency and tissue delivery capability. For instance, a study showed that exosomes purified from mesenchymal stem cell supernatants could be modified through electrostatic interactions with cationized pullulan, a polysaccharide. This biomaterial specifically targets hepatocyte asialoglycoprotein receptors, thereby improving the therapeutic potential of exosomes [127].
Surface engineering and modification techniques are essential for developing nanoparticles for specific applications and improving their performance in various fields. This could involve enhancing their stability, biocompatibility, dispersibility in a solvent, or targeting abilities in medical applications. Surface modification can also enable nanoparticles to carry specific molecules, such as drugs or targeting ligands, to enhance their functionality. It is further classified into two main types, as discussed below.
Surface coatings for improved stability and biocompatibility
Several methods have been employed to coat nanoparticles with different materials to enhance their biocompatibility. The surface coating of nanoparticles enhances their stability and decreases their adverse effects on cells, regardless of the surface charge of the nanoparticles [128]. Recent advancements in high-temperature synthesis techniques have enabled the production of high-quality iron oxide-based nanocrystals. These nanocrystals feature tunable magnetic properties influenced by their size and composition [129]. This control method has attracted increased interest in improving contrast effectiveness and understanding how these materials are distributed within organisms [130].
Nevertheless, concerns related to specific targeting within biological systems, how nanomaterials are distributed within organisms, and the potential for in vivo toxicity are significantly influenced by the stability of nanocrystals within intricate biological environments, their compatibility with living systems, and their size in fluid dynamics. These characteristics are directly influenced by one’s ability to consistently and efficiently integrate them with biological systems [21]. Coatings of nanoparticles have attracted special attention because they help release drugs, genes, and other bioactive substances in a controlled manner [131]. There are several common types of surface coatings for nanoparticles.
Polymer coatings: For the use of inorganic NPs in medicine, their surfaces must be easily mixed with water. Naturally, when nanoparticle surfaces repel water, they attract proteins and bind together, which is not good [132]. Some NPs can be made in an aqueous environment (i.e., gold NPs), while some inorganic NPs can be made in an aqueous environment and stabilised using hydrophobic molecules. Additional steps are taken to make them usable for biological purposes, such as coating or replacing these molecules with amphiphilic polymers [133]. Ligand exchange involves replacing existing surface ligands with new ones with a stronger binding affinity to the nanoparticle surface. In biological systems, this process often consists of substituting hydrophobic ligands with hydrophilic ones. To facilitate this exchange, the new ligands must possess a greater affinity for the surface than the existing ligands [52,134]. The binding strength between the attaching group and the surface of the NP primarily controls this phenomenon. The ligand affinity for the NP surface remains the critical determinant of the coating’s stability and resilience [133].
Surface modification techniques also incorporate extra functionalities into inorganic NPs. In biological systems, the configuration of the surface of NP is essential for influencing how the innate immune system identifies the NPs and controlling how the NPs are distributed within the body [135,136]. Hence, it becomes essential to protect NPs from fouling, a process characterised by the undesirable adhesion of macromolecules, microorganisms, or suspended particles onto the nanoparticle surface [137]. Hydrophilic polymers provide nanoparticle surfaces with a hydration layer to address this issue, which prevents fouling and enhances NP stability [138]. Polyethene glycol (PEG) continues to be the preferred choice for coating the surfaces of biomedical NPs because of its biocompatibility, straightforward application, and effective resistance to fouling [137,139]. PEG-based coatings can be tailored by adjusting parameters such as layer thickness, polymer architecture, and the chemical properties of the terminal groups. Increasing the molecular weight of PEG results in a thicker coating layer, while higher binding density promotes the formation of more extended PEG chains [135]. However, PEG and PEG binding derivatives onto specific substrates can pose challenges, increase the susceptibility to oxidation, and increase the potential to induce the production of antibodies against PEG (anti-PEG antibodies) [140]. Several alternative polymers, including Pluronics, polyacrylamides, polyacrylates, polyoxazolines, and polysaccharides, have been explored as potential substitutes for PEG in various applications [141].
The variations come from how water and the polymer molecules interact. Zwitterionic coatings connect with water through electrical charges, while PEG molecules use hydrogen bonds. The affinity for water can be adjusted by choosing specific opposing groups since the positive groups in zwitterionic coatings are usually quaternary ammonium. A more water-friendly nature increases when the acidity of the negative group decreases. The effectiveness of zwitterionic coatings on NPs in preventing fouling is linked to the strength of their hydration [142]. Zwitterionic polymers that are highly hydrated successfully avoid protein adsorption, while those with less robust hydration layers develop a protein corona. Apart from resisting proteins, these zwitterionic coatings also offer NPs the necessary stability for their use in biomedical applications [143].
Polymer coatings can be further enhanced by integrating responsiveness to specific stimuli. Stimulus-responsive polymers exhibit changes in their physical and chemical properties in response to environmental factors such as temperature, pH, or light [137]. Thermo- and pH-responsive poly-N-isopropylacrylamide (PNIPAm) is among the most studied stimuli-responsive polymers. Its solubility is determined by its critical solution temperature (CST), which can be tailored by modifying the polymer’s chain length and composition [137]. Moreover, polyalkyloxazolines (PAOXA) are thermoresponsive polymers that have garnered attention for their potential applications in biomedicine [144].
Lipid-based coatings: In recent decades, scientists have developed various nanomaterials with diverse shapes and compositions tailored explicitly for applications in biomedicine, with a specific focus on cancer diagnosis and treatment [145]. Lipids are naturally occurring components within cellular membranes that are crucial building blocks for their fundamental structure. The primary constituents of cell membranes are phospholipids, and their primary role is to act as a protective barrier, safeguarding the internal components of the cell from the external surroundings [146]. Lipids are promising candidates for various biomedical applications based on their biological functions. They offer a biocompatible protective layer on the surface of nanomaterials, mirroring their role in cellular membranes [147].
Inorganic NP cores frequently show instability in aqueous suspensions and may trigger cytotoxic effects. Consequently, numerous coating strategies have been proposed to enhance NP stability in aqueous solutions and their biocompatibility [148]. Among the array of components available for NP coatings, lipids, particularly phospholipids, have emerged as versatile molecular constituents for fabricating NP coatings tailored for biomedical applications [149]. Recent advancements in the synthesis of lipid coatings for NPs now enable the incorporation of a wide array of lipid molecules onto the nanoparticle surface. These lipid coatings can be combined with amphiphilic or hydrophobic drugs and active molecules for precise cell targeting [150]. The capacity to incorporate a variety of amphiphilic molecules with different functions into the nanoparticle coating’s outer layer provides additional adaptability to nanoparticle applications. Polymers, surfactants, and lipids commonly provide structural foundations for organic nanoparticle coatings [151]. The lipid class encompasses numerous molecules that exhibit a wide range of structures, but all share a common structural motif [152]. Usually, a lipid molecule can be divided into two distinct regions: hydrophobic and hydrophilic. In phospholipids, the hydrophobic section consists of one or two hydrocarbon chains with considerable variability in their length and degree of unsaturation. The composition of the hydrophilic region can also vary significantly, leading to lipids bearing negatively charged, positively charged, or zwitterionic groups, which are frequently encountered in nature [153].
Among all lipids, phospholipids possess a molecular structure and amphiphilic properties that align exceptionally well with the prerequisites for nanoparticle coating [154]. Zwitterionic phospholipids, such as phosphatidylcholine (PC), are commonly used for nanoparticle coatings. Charged molecules can be introduced on the NPS surface to enhance NPS stability, reducing aggregation. This can be achieved by combining zwitterionic phospholipids with anionic or cationic phospholipids. Alternatively, pegylated phospholipids create steric hindrance on the NPS surface, preventing aggregation due to their long PEG chains [155]. Since cellular membranes have negatively charged surfaces, it has been suggested that introducing a positive surface charge can enhance the uptake of biomedical nanodevices [156]. The inorganic core of NPs can serve various functions, such as drug-carrying (silica NPs), diagnostic (iron oxide NPs, gold, and QDs), therapeutic (iron oxide NPs, gold), and biosensing (silver NPs) functions. The specific function depends on the chemical composition of the inorganic component [157]. The lipid coating of NPs enhances their biocompatibility and regulates the release of active molecules from the nanoparticle core. Over time, lipid digestion and the gradual degradation of the nanoparticle core into nontoxic silicic acid and related byproducts contribute to the high biocompatibility of lipid-coated silica NPs [158].
Surface functionalization methods
Surface functionalization can be achieved through various methods. Ligand exchange or encapsulation was initially developed to transfer organically synthesised NPs into aqueous solutions. Conversely, wrapping and electrostatic conjugation are commonly employed in situ during the nanoparticle synthesis [159]. We will present an overview of the materials and techniques used for nanomaterial functionalization. The influence of these coatings on the biomedical application of nanomaterials will be discussed below.
Chemical conjugation techniques: Nanoparticles can be conjugated to proteins using two straightforward methods that avoid extensive chemical modifications or cloning: covalent bonding through cysteine-gold interactions or electrostatic attachment to the protein’s charged groups [160]. Chemical conjugation methods are commonly designed to target a protein’s amine, carboxyl, or thiol functional groups. Proteins have been conjugated with various molecules, including nucleic acids, peptides, other proteins, polymers, lipids, and nanoparticles [161]. Modifications typically target the side chain functionalities or the proteins’ C- and N-terminal moieties when binding other molecules to proteins. Amine groups in proteins, found at the N-terminus or lysine side chains, are positively charged and surface-accessible for conjugation. Overmodification of these groups can disrupt protein structure [162].
Thiols, unlike amines, are less common in proteins, offering a more specific conjugation target. However, they may not always be readily accessible on the protein surface due to disulfide bonds or steric hindrance. Thiol groups can be generated artificially by breaking disulfide bonds with reducing agents or introducing new thiols using thiolation reagents [163]. Carboxyl groups, located in aspartic and glutamic acid side chains or at the protein’s C-terminus, serve as a third target for conjugation. However, their natural reaction is challenging due to deprotonation by alkaline amine groups, requiring a carboxylic group activation reagent to facilitate amide formation through dehydration/condensation with an amine [164]. Copper (I)-catalysed azide-alkyne cycloaddition (CuAAC), the most used “click” reaction, is presented, including the introduction of the “click” concept, the conditions of copper (I) catalysis, the region selectivity, the nature of the catalysts and ligands, mechanistic features, experimental conditions and applications to organic synthesis and organic materials. CuAAC is an extensively recognised method for conjugating peptides with other peptides or polymers [165]. The conjugation effectiveness of CuAAC reactions can be enhanced by incorporating a copper catalyst and polymeric NPs [166].
Strain-promoted azide-alkyne cycloaddition (SPAAC) leverages cyclic alkynes’ inherent high ring strain to facilitate a chemically region-selective click reaction without additional catalytic assistance. Commonly referred to as the copper-free click reaction, it is a modified click chemistry approach that eliminates the requirement for copper, reducing agents, and accelerators [167]. SPARC enhances efficiency by replacing a linear alkyne group with a cyclic analogue, taking advantage of the ring strain in cycloalkyne. This approach is frequently used for protein-NP conjugation and exhibits high efficiency, surpassing N-hydroxysuccinimide (NHS) chemistry [168]. “Carbodiimides are reactive agents that activate carboxyl groups for reaction with amine groups under gentle conditions. Commercially available carbodiimides include 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) and dicyclohexylcarbodiimide (DCC). They function by reacting with carboxylic acids to form an acylisourea intermediate, which is then displaced by primary amines, resulting in an amide bond that connects the two molecules [169]. Antigen-conjugated nanoparticles can trigger robust immune responses without additional immunopotentiators like TLR agonists. Nonetheless, many of these systems benefit from external adjuvants for optimal efficacy. However, NP-based vaccine delivery systems offer advantages over nonconjugated approaches [161].
Bioconjugation approaches: Bioconjugation is the chemical procedure of creating a stable covalent bond between a biomolecule and another molecule, facilitating their attachment. Bioconjugation of NPs serves various purposes, including preventing NP clustering, loading drug molecules onto NPs, binding DNA or RNA to NPs, attaching targeting antibodies, or applying a protective coating to shield NPs from chemical reactions [170]. The conjugation of nanomaterials to biological components is pivotal in effectively applying nanomaterials. This step is crucial due to the numerous potential responses upon interaction with the biological environment, especially during internalisation [171]. B cells, essential immune system components, produce and secrete antibodies, which are highly valuable in nanobiotechnology and nanomedicine. Antibodies function through a key-lock mechanism, binding specifically to antigens on the surface of target cells [172]. Antibodies utilise their key-lock mechanism to bind to surface antigens, triggering their uptake into target cells via endocytosis. The variable amino acid regions enable this selective recognition at the tips of the antibody’s Y-shaped structure, which attaches explicitly to their corresponding antigens [173].
Biologically active species such as aptamers [174] or cell-penetrating peptides (CPPs) are composed of nucleic acid or amino acid sequences that can recognise specific proteins on cell surfaces. While these biomolecules demonstrate lower selectivity for cell attachment than antibodies, aptamers, short oligonucleotide DNA or RNA sequences, possess a unique three-dimensional folded structure [174]. CPPs comprise short amino acid sequences facilitating cell attachment by forming effective electrostatic interactions with phosphate or sulfonate groups in the cell membrane. CPPs do not serve as recognition agents but significantly aid cell penetration and internalisation. In recent reports, aptamers or CPPs have been attached to the surface of nanocarriers through covalent binding [175,176].
Folic acid (FA) is a biologically active compound that prepares targeted nanobioconjugates (NBCs). Compared to antibodies, FA offers greater stability, reduced sensitivity to temperature changes, and is a more cost-effective option [177]. FA acts as a specific cell-targeting agent by binding to the folate receptor on cell surfaces. While this receptor is generally expressed at low levels in most tissues, it is significantly overexpressed in many cancer cells, making FA a widely used option for targeted drug delivery in cancer research [177,178]. Confocal microscopy analysis revealed that the CFM@SiO2/PVA/Au-CR cargo exhibited improved internalisation into bacterial cells, including Escherichia coli (E. coli) and Klebsiella pneumoniae (K. pneumoniae). Optical density measurements revealed that this nanobiocomposite (NBC) system demonstrated significant antimicrobial activity against K. pneumoniae and E. coli, achieving inhibition rates of 93.0 ± 1.5% and 86.8 ± 1.0%, respectively. A key strength of this study lies in the enhanced antibacterial efficacy observed at reduced drug dosages [179]. Antibodies, aptamers, CPPs, and FAs are biologically active compounds in engineered biological systems. The advantages of these systems are the use of cell-responsive components such as proteins and peptides, which regulate cellular behaviour with high specificity and affinity [180]. The ideal features of nanocomposite (NBC) systems can be classified into five key categories: (1) straightforward synthesis, preparation, and functionalization of nanoparticles and NBCs, along with efficient drug loading and targeted delivery; (2) stability and biocompatibility of NBCs in the absence of drugs; (3) real-time monitoring of drug delivery using drug-loaded NBCs; (4) capability for stimuli-responsive drug release; and (5) adjustable control over drug toxicity. This survey comprehensively examined NBC drug carriers, including natural materials, MOFs, MNPs, COFs, and micelles [180].
Noncovalent surface modification strategies: Conte and Longobardi proposed a noncovalent strategy to improve the functionality of polyester aminic NPs by utilising electrostatic or lipophilic interactions [181] between the NPs and a peptide modified with an anchoring group. They used an iNGRt peptide (CRNGR) targeting NRP-1, which is found in cancers, and linked it with either poly or Palm to modify poly(ε-caprolactone) NPs. iNGRt-PolyE is attached to cationic NPs through electrostatic interactions, while iNGRt-Palm forms NPs. Both peptides are bound to ~100 nm NPs with positive zeta potentials. These nanoparticles successfully entered NRP-1-overexpressing MDA-MB-231 breast cancer cells, varying cell localisation based on the peptide anchoring. When loaded with the lipophilic drug DTX, iNGRt-Palm nanoparticles exhibited dose-dependent cytotoxicity comparable to DTX in MDA-MB-231 cells while showing reduced toxicity toward NRP-1-negative MRC-5 fibroblasts. In a mouse model of triple-negative breast cancer, iNGRt-Palm nanoparticles demonstrated superior tolerance, anticancer efficacy, and survival rates compared to free DTX, highlighting their potential for use in various biodegradable nanoparticle systems for therapeutic and targeting applications [182].
The characterisation and assessment of biofunctionalized nanoparticles are crucial for biomedical applications. These nanoparticles were rigorously tested for stability, size, and biocompatibility before being used for targeted medication administration and imaging. To ensure their safety and efficiency, wide-ranging research has been conducted on their interactions with biological systems, including their effects on cellular absorption and toxicity. By fusing nanotechnology with biology, this multidisciplinary approach makes it easier to produce cutting-edge healthcare tools, such as nanomedicines and biosensors, which have the potential to revolutionise medical diagnostics and treatments.
Physicochemical characterization techniques
Physiochemical characterisation techniques offer insights into NPs’ physical and chemical properties, including shape, size, surface charge, and composition. Detailed explanations of each method are provided as follows:
Size, morphology and Zeta potential analysis: Precise measurement of nanoparticle size, shape, and surface charge is foundational to understanding their physicochemical behaviour, colloidal stability, and biological performance. Several complementary analytical techniques are routinely employed to comprehensively characterise these properties, ensuring accuracy and reproducibility in nanoparticle-based applications [183]. In the context of nanoparticles, size analysis refers to the measurement and characterisation of the dimensions of nanoparticles, while morphological analysis focuses on examining their shape and structural characteristics. These analyses provide a comprehensive understanding of a nanoparticle’s physical attributes, aiding in the design and optimisation of nanoparticles [184].
Dynamic Light Scattering (DLS) remains the most widely used technique for determining the hydrodynamic diameter and polydispersity index (PDI) of nanoparticles dispersed in liquids. DLS analyses fluctuations in scattered light intensity due to Brownian motion, yielding rapid size estimations for colloidal systems. While highly convenient for monodisperse and spherical particles, DLS may show limited accuracy for polydisperse or irregularly shaped nanostructures [185]. Recent advancements, such as multi-angle DLS and backscattering configurations, have improved resolution for complex formulations. To complement DLS, Nanoparticle Tracking Analysis (NTA) has emerged as a powerful tool for direct visualisation and tracking of individual nanoparticle trajectories. By capturing the Brownian motion of single particles under a microscope and analysing it frame-by-frame, NTA provides number-based size distributions and estimates of particle concentration, revealing heterogeneity often masked in DLS data [186]. This capability makes NTA particularly valuable for analysing biological vesicles such as exosomes and virus-like particles.
Transmission Electron Microscopy (TEM) and its advanced variant, Scanning Transmission Electron Microscopy (STEM), are essential for directly observing nanoparticle morphology, internal structure, and crystalline features at nanometer or atomic resolution. STEM, equipped with high-angle annular dark-field detectors, enables Z-contrast imaging, which helps differentiate elements within bimetallic or core-shell structures [187]. Cryo-TEM techniques allow visualisation of nanoparticles in their near-native hydrated state, minimising drying-induced artefacts. Scanning Electron Microscopy (SEM) and Field Emission Scanning Electron Microscopy (FE-SEM) provide detailed surface topography and three-dimensional morphological information. FE-SEM, which uses a field emission gun as the electron source, offers higher resolution and more explicit images of nanostructures than conventional SEM. Sample preparation must ensure minimal aggregation to accurately represent the size and surface texture [188,189]. Atomic Force Microscopy (AFM) is increasingly employed to examine nanoparticles under ambient or liquid conditions, producing high-resolution three-dimensional surface profiles without requiring vacuum conditions. AFM can measure particle height, surface roughness, and mechanical properties such as stiffness or adhesion, adding a layer of information that electron microscopy alone cannot provide [190]. Moreover, force spectroscopy modes allow probing of ligand-receptor interactions at the nanoscale.
Zeta potential analysis quantifies the electrostatic potential at the slipping plane of dispersed nanoparticles, serving as a key indicator of colloidal stability and surface charge properties. Laser Doppler Electrophoresis (LDE) is the standard method for this purpose. LDE provides the zeta potential value by measuring the electrophoretic mobility of particles in an electric field, indicating the degree of electrostatic repulsion between particles. Nanoparticles with high positive or negative zeta potential (typically above ±30 mV) tend to remain well-dispersed due to strong repulsive forces, whereas low zeta potential values promote aggregation [191].
Recent studies highlight that zeta potential is highly sensitive to environmental conditions such as pH, ionic strength, and the presence of biomolecules. For instance, the adsorption of proteins or surfactants can dramatically shift surface charge, influencing nanoparticle-cell interactions and circulation behaviour. Surface modification with stabilising polymers like polyethene glycol (PEG) or targeting ligands can tailor zeta potential to enhance biocompatibility and minimize nonspecific uptake by the mononuclear phagocyte system. The size and surface morphology of biofunctionalized T. vulgaris silver nanoparticles were examined using TEM and SEM. Additionally, an EDS (energy-dispersive spectroscopy) analytical system was used to investigate the appearance of the elemental silver signal [192]. The morphology and size of polymyxin B-capped AgNPs (PB-AgNPs) were determined by TEM [193]. The sizes and morphologies of the silver nanoparticles were determined via HR-TEM and FE-SEM using a 200 kV accelerated voltage and an ultrahigh-resolution pole piece [194]. X-ray diffraction patterns are frequently used to determine the crystal structure of synthesised silver nanoparticles [195]. AgNPs have many uses in various biomedical applications, including cancer treatment and wound healing. Researchers have suggested that the efficiency of NPs can further increase with the use of these particles for gene delivery. These stable silver nanoparticles were biofunctionalized with suitable DNA-binding properties for effective transfection with minimal toxicity. Researchers reported on the one-pot facile green synthesis of silver nanoparticles modified with chitosan-g-polyacrylamide and stabilised with polyethene glycol (PEG). The particle size of these NPs examined via DLS was, to some extent, greater than that determined via TEM, as DLS assesses the hydrodynamic diameter in contrast to TEM, which images the dried NPs [196].
Surface charge measurements are crucial for understanding the physicochemical properties of biofunctionalized NPs and how they interact with biological systems. Zeta potential measurements are widely used to characterise the NP surface charge because they provide information on the stability and overall surface of the NPs in liquid media. By dislocating citrate anions, gold nanoparticles were functionalized with amine compounds and formed some agglomerations because of electrostatic relations and hydrogen bonding. With the addition of PVP polymer compounds, these aggregations can be avoided during functionalization. DLS was used to analyse the size and stability of the Au NPs and their functionalization [197].
Assessment of stability and drug loading efficiency: The temporal stability of the NPs is assessed with DLS as follows: determine the nanoparticles’ stability by evaluating the NPs’ sizes in DMEM and dH2O. Repeat the size measurements in DMEM and DI water once/day for five days [198]. The zeta potential of synthesised NPs was about -12.6 mV, which suggests that the biofunctionalized TVAgNPs are more stable. The higher negative surface charge potential can support long-term stability and high dispersion [192].
An efficient and effective method was employed to synthesise biofunctionalized NPs using polymyxin B (an AMP). Polymyxin B is a well-known cationic, nonribosomal peptide shown to have antibacterial effects by interacting with the lipid A portion of the LPS (lipopolysaccharide of the outer membrane of gram-negative bacteria. PBSNPs were maintained at 37 °C for stability analysis, and samples were taken at various intervals to check for polymyxin B leaching into the solution. Additionally, the stability of PBSNPs kept at 4 °C was assessed by monitoring the antibacterial activity over one month [193]. Evaluating the capability of biofunctionalized NPs to load drugs is one of the most essential steps in creating specialised drug delivery systems. Zhang, et al. (2022) reported the development of a nanoparticle designed to target inflammation sites and selectively release PMB at locations with gram-negative bacteria. Moreover, in the study of Gounani, et al. (2018), bare and surface-modified mesoporous silica nanoparticles (MSNPs) are used as adsorbents for polymyxin B to improve its therapeutic properties. Biocompatibility assessments showed that loading polymyxin B onto mesoporous silica nanoparticles (MSNs) reduced the drug’s cytotoxic effects by lowering ROS generation. These findings suggest that drug formulations using adsorption onto MSNs could mitigate the adverse effects of antibiotics like polymyxin B while retaining their antimicrobial efficacy [199,200].
In vitro and in vivo evaluation of therapeutic efficacy and imaging capabilities
Silica NPs are among the most promising nanocarriers and have many considerations. Additionally, the FDA has approved silica NPs for use in biomedical applications [201,202]. MSNPs with significant physicochemical characteristics have gained massive attention among the mesoporous family for research in various domains, including catalysis and drug delivery [203]. Mesoporous morphology provides them with a wide surface area, easy surface functionalization, customizable pore size, and highly organised channels for therapeutic chemicals loading. Make them suitable as a nanocontainer for cancer treatment, tissue engineering, and theranostic applications [204]. The unique properties of MSNPs make them a perfect candidate for biomedical applications [205].
A sequence of potent anticancer chelators, dipyridyl thiosemicarbazone chelators, has been demonstrated to defeat MDR in malignant cells. These chelators, led by Dp44mT, bind to copper and iron to make redox-active complexes, permeate the lysosome membrane, and defeat P-glycoprotein-mediated drug resistance in cancer cells [206]. Dp44mT has been shown to upregulate the powerful metastasis suppressor N-myc downstream-regulated gene-1 (NDRG1), which inhibits the epithelial-to-mesenchymal transition, suppresses oncogenic signalling, and reduces tumour cell migration and metastasis in vivo. Thus, this chelator has enormous potential as a chemotherapeutic drug for treating untreatable glioma and aggressive tumours. Some earlier research has revealed the excellent ability of this chelator to fight glioma tumours, with IC50 values (greater than 150 mM) significantly lower than those reported for other frequently used anticancer agents like TMZ and DOX (2-33 M range) [207].
In vivo imaging procedures are necessary not only to recognise the tumour heterogeneity and diagnose cancer but also to monitor constant changes in the state of the patient’s disease. As a result, several imaging tools, including PET (positron emission tomography), optical fluorescence, SPECT (single photon emission computed tomography), PAI (photoacoustic imaging), MRI (magnetic resonance imaging), CT (computed tomography) and US (ultrasound) have been developed and used for clinical and preclinical purposes [21,208]. In addition, Chang et al. proved that the nanocomposite hydrogel functional wound dressing prepared by combining AgNPs and PTT could alleviate inflammation and promote the healing of infected skin wounds [209].
Cell-based assays and cytotoxicity studies: Biochemical or cellular functions can be measured using a variety of cell-based assays. These in vitro assays can be used to examine the toxicity of test substances or how they affect different cell functions, such as adipogenesis, apoptosis, angiogenesis, senescence, and viability. Researchers have demonstrated interactions between nanoparticles and cells or biological systems, referred to as bio-nano interactions [210,211]. A deep understanding of bio-nano interactions is essential for synthesising nanomaterials, their nanotoxicity in the environment, and their biological applications. Nanomaterials can be easily engineered to interact with cells in specific ways, making it possible to employ them as molecular probes for imaging, drug delivery carriers, or vectors. Researchers have focused on assays utilised to examine the bio-nano interactions between mammalian cells and nanomaterials [212]. Researchers identified the appropriate test for determining Fe3O4 nanoparticles in hMSCs (human mesenchymal stem cells) following short-term exposure using various viability tests, such as MTT, NRU, and TB assays, in parallel with cell morphology analysis for cross-checking. These findings conflict with morphological investigations showing decreased loss of monolayer features, cell density, and morphological changes in the presence of ≥50 μg/ml Fe3O4 NPs. TB treatment resulted in a considerable effect on hMSCs (25%) at concentrations of ≥10 μg/ml Fe3O4NPs, with a maximal outcome (45%) at 300 μg/ml after 24 hours, which was aggravated after 48 hours. The results indicated that spectrophotometric assays do not apply to hMSC culture conditions. However, TB is a reliable approach for assessing cell viability following Fe3O4NP revelation in this model [213]. A research group found that assays focused on specific cell functions were more sensitive than traditional tests, such as proliferation, ROD, and LDH, in detecting damage from TiO2 exposure in ADSCs. Despite no detectable damage in conventional tests, they observed significant changes in ECM protein secretion and reduced angiogenesis in cultures exposed to low concentrations of TiO2 nanoparticles. These changes could impair two vital functions of ADSCs and pose potential risks to human health. Moreover, TiO2 exposure led to a concentration-dependent increase in ADSC cytotoxicity by elevating ROS levels and extracellular LDH, disrupting ECM protein production, and reducing wound healing capacity and differentiation [214].
Animal models and biodistribution analysis: For the first time, researchers have considered the safety, pharmacokinetics, and biodistribution of dextran-coated gold nanoparticles produced via intravenous laser synthesis in a small animal model. Studies indicated that these nanoparticles predominantly accumulated in the spleen and liver without causing liver or kidney damage, as confirmed by stable biochemical markers and histological tissue analysis. The safety evaluation included observing expected animal behaviour and the absence of acute or chronic toxicity in major organs. Although some residual accumulation was observed, laser-produced nanoparticles were generally safe and held significant potential for biomedical applications such as drug delivery, contrast imaging, and therapeutic sensitisation [215].
In a related study, DiR was used as a nanoparticle tracer to examine the in vivo distribution of polymeric nanoparticles in mice through NIR fluorescence whole-body imaging. Polymerised tannic acid (pTA) served as a sticky intermediary layer, enabling the surface modification of PLGA nanoparticles with either folate-conjugated polyethene glycol (pEol) or methoxy-terminated polyethene glycol (PEG). Mice with folate receptor-overexpressing tumours were intravenously injected with pEol-modified PLGA nanoparticles loaded with DiR to evaluate folate-dependent nanoparticle distribution. Interestingly, the fluorescence distribution patterns were influenced more significantly by the DiR loading content than by the surface ligand, leading to contrasting conclusions despite using the same nanoparticle type. The study also identified two distinct behaviours, quenching and saturation of DiR-loaded PLGA nanoparticles, which could affect in vivo fluorescence kinetics and intensity profiles [216].
Scientists have compared the biodistribution of silicon NPs and carbon dots with small sizes under 5 nm. To obtain accurate information about biodistribution via PET, the particles were outfitted with a NOTA tag for labelling with 64Cu as a positron emitter. Experiments in small animals using tumour-bearing mice and Wistar rats demonstrated that all the particles were quickly eliminated from the body. According to experiments in small animals using tumour-bearing mice, all the particles were removed rapidly from the body. The surface characteristics of the described particles mainly determined their behaviour both in vivo and in vitro. The surface coverage of the amine groups and the amount of surfactant that remained after their production and purification were related to their toxicity [217].
Imaging techniques for in vivo visualisation and tracking: City imaging is clinically appropriate, allowing high-resolution, three-dimensional anatomical imaging and good tissue penetration. Accumulating evidence has shown the viability of CT cells tracked with gold nanoparticles in vivo. This concept offers a variety of benefits, such as the capacity to conduct a longitudinal investigation in parallel with anatomical imaging and quantify even relatively small cell numbers. The long-term viability of cells and the uncertainty about the retention of particles in cells over time are the fundamental drawbacks of CT cell tracking. Long-term therapeutic effects were observed in various animal models treated with AuNP-loaded cells, supporting long-term viability and functionality; however, further technological developments are required to obtain reliable data over more extended periods [218]. Over the last ten years, the use of molecular imaging techniques for tracking stem cell behaviour in vivo has significantly increased, and these techniques have shown potential in both clinical and preclinical trials. In recent years, various attempts have been made to directly label stem cells using a variety of contrast agents, including gold nanoparticles, fluorescent dyes, and magnetic particles [219]. For this purpose, imaging agents have been delivered to stem cells using a variety of NPs, which can enter the cells by active or passive transport and are trapped inside the cells. However, heterogeneous stem cells with a low endocytic capability do not respond well to the internalisation of NPs. These factors make it difficult to control the efficiency of NP labelling actively.
Biofunctionalisation of nanoparticles has evolved into a central paradigm for addressing the intrinsic limitations of conventional therapeutic and diagnostic systems. Through rational surface modification with antibodies, peptides, aptamers, and small-molecule ligands, nanoplatforms now exhibit markedly improved targeting precision, biological compatibility, and functional versatility. These advances have enabled sophisticated applications spanning targeted drug delivery, molecular imaging, biosensing, and integrated theranostic systems. The synergistic integration of ligand-mediated recognition with stimuli-responsive mechanisms further expands the capacity of nanoparticles to operate effectively within complex and dynamic biological microenvironments. Such multifunctional architectures represent a foundational step toward precision medicine, where diagnosis, treatment, and monitoring converge within unified nanoscale systems.
Despite substantial progress, translational barriers remain significant. Challenges involving scalable and reproducible synthesis, preservation of surface integrity, long-term in vivo stability, immunogenicity risks, and chronic toxicity must be systematically resolved. Standardised safety evaluation frameworks, predictive biodistribution modelling, and regulatory harmonisation are essential for clinical maturation. Looking forward, artificial intelligence (AI) is poised to redefine nanoparticle engineering. Machine learning models can optimise physicochemical parameters, predict nano–bio interactions, and guide ligand selection using multidimensional datasets. AI-assisted design enables inverse engineering approaches, accelerating the discovery of nanoparticles with predefined pharmacokinetic, targeting, and safety profiles. Coupled with high-throughput screening and computational simulations, these tools significantly reduce experimental iteration cycles.
Simultaneously, biomimetic platforms represent a transformative frontier. Cell membrane-coated nanoparticles, exosome-inspired vesicles, protein-corona engineering, and bioinspired adaptive materials offer enhanced immune evasion, prolonged circulation, and superior biological interfacing. These systems emulate endogenous transport pathways, improving targeting fidelity and therapeutic efficiency. The fusion of biomimetic strategies with AI-driven optimisation may yield self-adaptive, context-aware nanoplatforms capable of dynamic responses to physiological signals. Collectively, the convergence of advanced biofunctionalisation, AI-guided design, and biomimetic engineering establishes a robust framework for next-generation nanomedicine. Continued interdisciplinary integration will be critical for translating multifunctional nanoparticles from experimental constructs into clinically reliable technologies.
- Haleem A, Javaid M, Singh RP, Rab S, Suman R. Applications of nanotechnology in medical field: a brief review. Global Health Journal. 2023;7(2):70-77. Available from: https://doi.org/10.1016/j.glohj.2023.02.008
- Madamsetty VS, Paul MK, Mukherjee A, Mukherjee S. Functionalization of nanomaterials and their application in melanoma cancer theranostics. ACS Biomaterials Science & Engineering. 2019;6(1):167-181. Available from: https://doi.org/10.1021/acsbiomaterials.9b01426
- Kiani MN, Khaliq H, Abubakar M, Rafique M. Advancing the potential of nanoparticles for cancer detection and precision therapeutics. Medical Oncology. 2025;42(7):1-32. Available from: https://doi.org/10.1007/s12032-025-02782-6
- Zubair M, Riaz M, Kiani MN, Aslam HM. Application of nanotechnology for targeted drug delivery and nontoxicity. International Journal of General Practice Nursing. 2024;2(2):57-67. Available from: https://www.researchgate.net/publication/382364082_Application_of_Nanotechnology_for_Targeted_Drug_Delivery_and_Nontoxicity
- Navya P, Kaphle A, Srinivas SP, Bhargava SK, Rotello VM, Daima HK. Current trends and challenges in cancer management and therapy using designer nanomaterials. Nano Convergence. 2019;6(1):1-30. Available from: https://doi.org/10.1186/s40580-019-0193-2
- Liu R, Luo C, Pang Z, Zhang J, Ruan S, Wu M. Advances of nanoparticles as drug delivery systems for disease diagnosis and treatment. Chinese Chemical Letters. 2023;34(2):107518. Available from: https://doi.org/10.1016/j.cclet.2022.05.032
- Kiani MN, Butt MS, Gul IH, Saleem M, Irfan M, Baluch AH, et al. Synthesis and characterisation of cobalt-doped ferrites for biomedical applications. ACS Omega. 2023;8(4):3755-3761. Available from: https://doi.org/10.1021/acsomega.2c05226
- Suhag D, Kaushik S, Taxak VB. Theranostics: Combining Diagnosis and Therapy, in Handbook of Biomaterials for Medical Applications, Volume 1: Fundamentals. Springer. 2024;271-295. Available from: https://www.researchgate.net/publication/382505478_Theranostics_Combining_Diagnosis_and_Therapy
- Altinbasak I, Alp Y, Sanyal R, Sanyal A. Theranostic nanogels: multifunctional agents for simultaneous therapeutic delivery and diagnostic imaging. Nanoscale. 2024;16(29):14033-14056. Available from: https://pubs.rsc.org/en/content/articlelanding/2024/nr/d4nr01423e
- Khan AU, Ilyas M, Zamel D, Khan S. Bio-inspired fabrication of zinc oxide nanoparticles: Insight into biomedical applications. Annals of Advances in Chemistry. 2022;6(1):023-037. Available from: https://doi.org/10.29328/journal.aac.1001028
- Al-Thani AN, Jan AG, Abbas M, Geetha M, Sadasivuni KK. Nanoparticles in cancer theragnostic and drug delivery: A comprehensive review. Life Sciences. 2024;352:122899. Available from: https://doi.org/10.1016/j.lfs.2024.122899
- Uniyal S, Choudhary K, Sachdev S, Kumar S. Nano-bio fusion: Advancing biomedical applications and biosensing with functional nanomaterials. Optics & Laser Technology. 2024;168:109938. Available from: https://doi.org/10.1016/j.optlastec.2023.109938
- Zamel D, Khan AU, Elmasry SA, Elsayed ASS, Khan S, Muhammad M, et al. Biofunctionalisation of Carbon Nanostructures, in Handbook of Functionalized Carbon Nanostructures: From Synthesis Methods to Applications. Springer. 2024;1215-1251. Available from: https://link.springer.com/rwe/10.1007/978-3-031-32150-4_36
- Rajaei M, Rashedi H, Yazdian F, Pourmadadi M, Rahdar A, Pandey S. Chitosan Nanocarriers: Pioneering Encapsulation and Targeted Delivery of 5-Fluorouracil-A Comprehensive Review. European Journal of Medicinal Chemistry Reports. 2024;100172. Available from: https://doi.org/10.1016/j.ejmcr.2024.100172
- Tavares TD, Antunes JC, Ferreira F, Felgueiras HP. Biofunctionalisation of natural fibre-reinforced biocomposites for biomedical applications. Biomolecules. 2020;10(1):148. Available from: https://doi.org/10.3390/biom10010148
- Di S, Qian Y, Wang L, Li ZL. Biofunctionalisation of graphene and its two-dimensional analogues and synthesis of biomimetic materials: A review. Journal of Materials Science. 2022;1-29. Available from: https://www.researchgate.net/publication/357553795_Biofunctionalization_of_graphene_and_its_two-dimensional_analogues_and_synthesis_of_biomimetic_materials_a_review
- Kumar KB, Rajitha A, Rao AK, Alam K, Albawi A, Sethi G. SMART Materials for Biomedical Applications: Advancements and Challenges. E3S Web of Conferences. 2023. Available from: https://doi.org/10.1051/e3sconf/202343001133
- Khan AU, Ilyas M, Zamel D, Khan S, Ahmad A, Kaneez F. Bio-inspired fabrication of zinc oxide nanoparticles: Insight into biomedical applications. Advance Chemistry Journal. 2022. Available from: https://www.advancechemjournal.com/index.php/aac/article/view/aac-aid1028
- Khan AU, Waris A, Zamel D, Mbayachi VB, Muhaymin A, Safdar A, Shahid Z, et al. Biological and Chemical Synthesis of Nanoparticles. In: Chemical Physics of Polymer Nanocomposites: Processing, Morphology, Structure, Thermodynamics, Rheology. Wiley. 2024;1:15-44. Available from: https://research-information.bris.ac.uk/en/publications/biological-and-chemical-synthesis-of-nanoparticles/
- Niu B, Yang K, Lawrence B, Ge H. Transient Ligand‐Enabled Transition Metal-Catalysed C− H Functionalization. ChemSusChem. 2019;12(13):2955-2969. Available from: https://doi.org/10.1002/cssc.201900151
- Ahmad F, Salem-Bekhit MM, Khan F, Alshehri S, Khan A, Ghoneim MM. Unique properties of surface-functionalized nanoparticles for bio-application: functionalization mechanisms and importance in application. Nanomaterials. 2022;12(8):1333. Available from: https://doi.org/10.3390/nano12081333
- Jing X, Zhang Y, Li M, Zuo X, Fan C, Junhua et al. Surface engineering of colloidal nanoparticles. Materials Horizons. 2023;10(4):1185-1209. Available from: https://pubs.rsc.org/en/content/articlelanding/2023/mh/d2mh01512a
- Bandyopadhyay A, Das T, Nandy S, Sahib S, Preetam S, Gopalakrishnan AV, et al. Ligand-based active targeting strategies for cancer theranostics. Naunyn-Schmiedeberg's Archives of Pharmacology. 2023;396(12):1-25. Available from: https://doi.org/10.1007/s00210-023-02612-4
- Yoo J, Park C, Yi G, Lee D, Koo H. Active Targeting Strategies Using Biological Ligands for Nanoparticle Drug Delivery Systems. Cancers. 2019;11(5):640. Available from: https://doi.org/10.3390/cancers11050640
- Ahmad A, Imran M, Ahsan H. Biomarkers as Biomedical Bioindicators: Approaches and Techniques for the Detection, Analysis, and Validation of Novel Biomarkers of Diseases. Pharmaceutics. 2023;15(6). Available from: https://doi.org/10.3390/pharmaceutics15061630
- Sharma A, Bhargava P, Prasad R, Choudhary DK. Nanosensors in Healthcare Diagnostics. Elsevier. 2024. Available from: https://shop.elsevier.com/books/nanosensors-in-healthcare-diagnostics/sharma/978-0-443-19129-9
- Bayat P, Nosrati R, Alibolandi M, Rafatpanah H, Abnous K, Khedri M, et al. SELEX methods on the road to protein targeting with nucleic acid aptamers. Biochimie. 2018;154:132-155. Available from: https://doi.org/10.1016/j.biochi.2018.09.001
- Ramesh M, Janani R, Deepa C, Rajeshkumar L. Nanotechnology-Enabled Biosensors: A Review of Fundamentals, Design Principles, Materials, and Applications. Biosensors. 2023;13(1):40. Available from: https://doi.org/10.3390/bios13010040
- Johnsen KB, Burkhart A, Thomsen LB, Andresen TL, Moos T. Targeting the transferrin receptor for brain drug delivery. Progress in Neurobiology. 2019;181:101665. Available from: https://doi.org/10.1016/j.pneurobio.2019.101665
- Aydin S, Emre E, Ugur K, Aydin MA, Sahin I, Cinar V, et al. An overview of ELISA: a review and update on best laboratory practices for quantifying peptides and proteins in biological fluids. Journal of International Medical Research. 2025;53(2):3000605251315913. Available from: https://doi.org/10.1177/03000605251315913
- Bae Y, Jang DG, Eom S, Park TJ, Kang S. HRP-conjugated plug-and-playable IgG-binding nanobodies as secondary antibody mimics in immunoassays. Sens Actuators B Chem. 2020;320:128312. Available from: https://doi.org/10.1016/j.snb.2020.128312
- Spada A, Gerber-Lemaire S. Surface functionalization of nanocarriers with anti-EGFR ligands for cancer active targeting. Nanomaterials (Basel). 2025;15(3):158. Available from: https://doi.org/10.3390/nano15030158
- Huang L, Huang Z, Zhang Y, Lin C, Zhao Z, Li R, et al. Advances in targeted delivery of mRNA into immune cells for enhanced cancer therapy. Theranostics. 2024;14(14):5528. Available from: https://doi.org/10.7150/thno.93745
- Chehelgerdi M, Allela OQB, Pecho RDC, Jayasankar N, Rao DP, Thamaraikani T, Vasanthan M, et al. Progressing nanotechnology to improve targeted cancer treatment: overcoming hurdles in its clinical implementation. Mol Cancer. 2023;22(1):169. Available from: https://doi.org/10.1186/s12943-023-01865-0
- Guo YY, Huang L, Zhang ZP, Fu DH. Strategies for precise engineering and conjugation of antibody targeted-nanoparticles for cancer therapy. Curr Med Sci. 2020;40(3):463-473. Available from: https://doi.org/10.1007/s11596-020-2200-6
- Hauser-Kawaguchi A, Luyt LG, Turley E. Design of peptide mimetics to block pro-inflammatory functions of HA fragments. Matrix Biol. 2019;78:346-356. Available from: https://doi.org/10.1016/j.matbio.2018.01.021
- Sis MJ, Webber MJ. Drug delivery with designed peptide assemblies. Trends Pharmacol Sci. 2019;40(10):747-762. Available from: https://doi.org/10.1016/j.tips.2019.08.003
- Sadraeian M, Maleki R, Moraghebi M, Bahrami A. Phage display technology in biomarker identification with emphasis on non-cancerous diseases. Molecules. 2024;29(13):3002. Available from: https://doi.org/10.3390/molecules29133002
- Shang S, Li X, Wang H, Zhou Y, Pang K, Li P. Targeted therapy of kidney disease with nanoparticle drug delivery materials. Bioact Mater. 2024;37:206-221. Available from: https://doi.org/10.1016/j.bioactmat.2024.03.014
- Liu N, Li M, Xie F, Lv J, Gao X, Zhang H, et al. Efficacy of mimetic viral dynein binding peptide binding nanoparticles in blood-brain barrier model. J Drug Deliv Sci Technol. 2022;74:103523. Available from: https://doi.org/10.1016/j.jddst.2022.103523
- Singh R, Srinivas SP, Kumawat M, Daima HK. Ligand-based surface engineering of nanomaterials: trends, challenges, and biomedical perspectives. OpenNano. 2024;15:100194. Available from: https://doi.org/10.1016/j.onano.2023.100194c
- Zhou W, Gao X, Liu D, Chen X. Gold nanoparticles for in vitro diagnostics. Chem Rev. 2015;115(19):10575-10636. Available from: https://doi.org/10.1021/acs.chemrev.5b00100
- Leung KK, Schaefer K, Lin Z, Yao Z, Wells JA. Engineered proteins and chemical tools to probe the cell surface proteome. Chem Rev. 2025;125(8):4069-4110. Available from: https://doi.org/10.1021/acs.chemrev.4c00554
- Radaeva M, Ton AT, Hsing M, Ban F, Cherkasov A. Drugging the ‘undruggable’: therapeutic targeting of protein-DNA interactions with the use of computer-aided drug discovery methods. Drug Discov Today. 2021;26(11):2660-2679. Available from: https://doi.org/10.1016/j.drudis.2021.07.018
- Roberts TC, Langer R, Wood MJA. Advances in oligonucleotide drug delivery. Nat Rev Drug Discov. 2020;19(10):673-694. Available from: https://doi.org/10.1038/s41573-020-0075-7
- Zhang Y, Tu J, Wang D, Zhu H, Maity SK, Qu X, et al. Programmable and multifunctional DNA-based materials for biomedical applications. Adv Mater. 2018;30(24):1703658. Available from: https://doi.org/10.1002/adma.201703658
- Tan J, Yang N, Hu Z, Su J, Zhong J, Yang Y, et al. Aptamer-functionalized fluorescent silica nanoparticles for highly sensitive detection of leukaemia cells. Nanoscale Res Lett. 2016;11(1):1-8. Available from: https://doi.org/10.1186/s11671-016-1512-8
- Kadam US, Hong JC. Advances in aptameric biosensors designed to detect toxic contaminants from food, water, human fluids, and the environment. Trends Environ Anal Chem. 2022;36:e00184. Available from: https://doi.org/10.1016/j.teac.2022.e00184
- Zhang W, Taheri-Ledari R, Ganjali F, Afruzi FH, Hajizadeh Z, Saeidirad M, et al. Nanoscale bioconjugates: a review of the structural attributes of drug-loaded nanocarrier conjugates for selective cancer therapy. Heliyon. 2022;8(6):e09577. Available from: https://doi.org/10.1016/j.heliyon.2022.e09577
- Śmiłowicz D, Slootweg JC, Metzler-Nolte N. Bioconjugation of cyclometalated gold (III) lipoic acid fragments to linear and cyclic breast cancer targeting peptides. Mol Pharm. 2019;16(11):4572-4581. Available from: https://doi.org/10.1021/acs.molpharmaceut.9b00695
- António JP, Russo R, Carvalho CP, Cal PMSD, Gois PMP. Boronic acids as building blocks for the construction of therapeutically useful bioconjugates. Chemical Society Reviews. 2019;48(13):3513-3536. Available from: https://doi.org/10.1039/C9CS00184K
- Heuer-Jungemann A, Feliu N, Bakaimi I, Hamaly M, Alkilany A, Chakraborty I, et al. The role of ligands in the chemical synthesis and applications of inorganic nanoparticles. Chemical Reviews. 2019;119(8):4819-4880. Available from: https://doi.org/10.1021/acs.chemrev.8b00733
- Mu J, Meng X, Chen L, Lu Z, Mou Q, Li X, et al. Highly stable and biocompatible W18O49@PEG-PCL hybrid nanospheres combining CT imaging and cancer photothermal therapy. RSC Advances. 2017;7(18):10692-10699. Available from: https://pubs.rsc.org/en/content/articlelanding/2017/ra/c6ra28161c
- Sanità G, Carrese B, Lamberti A. Nanoparticle surface functionalization: How to improve biocompatibility and cellular internalisation. Frontiers in Molecular Biosciences. 2020;7:587012. Available from: https://doi.org/10.3389/fmolb.2020.587012
- Kostiv U, Patsula V, Šlouf M, Pongrac IM, Škokić S, Radmilović MD, et al. Physico-chemical characteristics, biocompatibility, and MRI applicability of novel monodisperse PEG-modified magnetic Fe3O4&SiO2 core–shell nanoparticles. RSC Advances. 2017;7(15):8786-8797. Available from: https://doi.org/10.1039/C7RA00224F
- Sanità G, Carrese B, Lamberti A. Nanoparticle surface functionalization: How to improve biocompatibility and cellular internalisation. Front Mol Biosci. 2020;7:587012. Available from: https://doi.org/10.3389/fmolb.2020.587012
- Chauhan P, Ragendu V, Kumar M, Molla R, Mishra SD, Basa S, et al. Chemical technology principles for selective bioconjugation of proteins and antibodies. Chemical Society Reviews. 2024;53(1):380-449. Available from: https://doi.org/10.1039/d3cs00715d
- Bednarek C, Schepers U, Thomas F, Bräse S. Bioconjugation in materials science. Advanced Functional Materials. 2024;34(20):2303613. Available from: https://doi.org/10.1002/adfm.202303613
- Eras A, Castillo D, Suárez M, Vispo NS, Albericio F, Rodriguez H. Chemical conjugation in drug delivery systems. Frontiers in Chemistry. 2022;10:889083. Available from: https://doi.org/10.3389/fchem.2022.889083
- Rosero WAA, Barbezan AB, de Souza CD, Rostelato MECM. Review of advances in coating and functionalization of gold nanoparticles: From theory to biomedical application. Pharmaceutics. 2024;16(2):255. Available from: https://doi.org/10.3390/pharmaceutics16020255
- Lei Z, Chen H, Huang S, Wayment LJ, Xu Q, Zhang W. New advances in covalent network polymers via dynamic covalent chemistry. Chemical Reviews. 2024;124(12):7829-7906. Available from: https://doi.org/10.1021/acs.chemrev.3c00926
- Szwed-Georgiou A, Płociński P, Kupikowska-Stobba B, Urbaniak MM, Rusek-Wala P, Szustakiewicz K, et al. Bioactive materials for bone regeneration: biomolecules and delivery systems. ACS Biomaterials Science & Engineering. 2023;9(9):5222-5254. Available from: https://doi.org/10.1021/acsbiomaterials.3c00609
- Cerkezi S, Nakova M, Gorgoski I, Ferati K, Bexheti-Ferati A, Palermo A, et al. The role of sulfhydryl (thiols) groups in oral and periodontal diseases. Biomedicines. 2024;12(4):882. Available from: https://doi.org/10.3390/biomedicines12040882
- Kumar S, Kumar A, Sharma D, Das P. Free amine, hydroxyl and sulfhydryl directed C−H functionalization and annulation: Application to heterocycle synthesis. The Chemical Record. 2022;22(2):e202100171. Available from: https://doi.org/10.1002/tcr.202100171
- Worch JC, Stubbs CJ, Price MJ, Dove AP. Click nucleophilic conjugate additions to activated alkynes: Exploring thiol-yne, amino-yne, and hydroxyl-yne reactions from (bio)organic to polymer chemistry. Chemical Reviews. 2021;121(12):6744-6776. Available from: https://doi.org/10.1021/acs.chemrev.0c01076
- Monga A, Nandini D. Synthetic access to thiols: A review. Journal of Chemical Sciences. 2024;136(4):67. Available from: https://www.ias.ac.in/public/Volumes/jcsc/136/00/0067.pdf
- Kaymaz SV, Nobar HM, Sarıgül H, Soylukan C, Akyüz L, Yüce M. Nanomaterial surface modification toolkit: Principles, components, recipes, and applications. Advances in Colloid and Interface Science. 2023;322:103035. Available from: https://doi.org/10.1016/j.cis.2023.103035
- Sahle FF, Gulfam M, Lowe TL. Design strategies for physical-stimuli-responsive programmable nanotherapeutics. Drug Discovery Today. 2018;23(5):992-1006. Available from: https://doi.org/10.1016/j.drudis.2018.04.003
- Li F, Zhang F, Yi X, Quan LL, Yang X, Yin C, et al. Proline hydroxylase 2 (PHD2) promotes brown adipose thermogenesis by enhancing the hydroxylation of UCP1. Molecular Metabolism. 2023;73:101747. Available from: https://doi.org/10.1016/j.molmet.2023.101747
- Deirram N, Zhang C, Kermaniyan SS, Johnston APR, Such GK. pH-responsive polymer nanoparticles for drug delivery. Macromolecular Rapid Communications. 2019;40(10):1800917. Available from: https://doi.org/10.1002/marc.201800917
- Li L, Yang Y, Lv Y, Yin P, Lei T. Porous calcite CaCO3 microspheres: Preparation, characterisation, and release behaviour as doxorubicin carrier. Colloids and Surfaces B: Biointerfaces. 2020;186:110720. Available from: https://doi.org/10.1016/j.colsurfb.2019.110720
- Beach MA, Nayanathara U, Gao Y, Zhang C, Xiong Y, Wang Y, et al. Polymeric nanoparticles for drug delivery. Chemical Reviews. 2024;124(9):5505-5616. Available from: https://doi.org/10.1021/acs.chemrev.3c00705
- Ow V, Lin Q, Wong JHM, Sim B, Tan YL, Leow Y, et al. Understanding the interplay between pH and charges for theranostic nanomaterials. Nanoscale. 2025;17(12):6960-6980. Available from: https://doi.org/10.1039/D4NR03706E
- Ellis E, Zhang K, Lin Q, Ye E, Poma A, Battaglia G, et al. Biocompatible pH-responsive nanoparticles with a core-anchored multilayer shell of triblock copolymers for enhanced cancer therapy. Journal of Materials Chemistry B. 2017;5(23):4421-4425. Available from: https://doi.org/10.1039/c7tb00654c
- Wang Z, Luo M, Mao C, Wei Q, Zhao T, Li Y, et al. A redox-activatable fluorescent sensor for the high-throughput quantification of cytosolic delivery of macromolecules. Angewandte Chemie. 2017;129(5):1339-1343. Available from: https://doi.org/10.1002/anie.201610302
- Gulfam M, Sahle FF, Lowe TL. Design strategies for chemical-stimuli-responsive programmable nanotherapeutics. Drug Discovery Today. 2019;24(1):129-147. Available from: https://doi.org/10.1016/j.drudis.2018.09.019
- Jia R, Du J, Cao L, Feng W, He Q, Xu P, et al. Application of transcriptome analysis to understand the adverse effects of hydrogen peroxide exposure on brain function in common carp (Cyprinus carpio). Environmental Pollution. 2021;286:117240. Available from: https://doi.org/10.1016/j.envpol.2021.117240
- Rahikkala A, Aseyev V, Tenhu H, Kauppinen EI, Raula J. Thermoresponsive nanoparticles of self-assembled block copolymers as potential carriers for drug delivery and diagnostics. Biomacromolecules. 2015;16(9):2750-2756. Available from: https://doi.org/10.1021/acs.biomac.5b00690
- Huang H, Qi X, Chen Y, Wu Z. Thermo-sensitive hydrogels for delivering biotherapeutic molecules: A review. Saudi Pharmaceutical Journal. 2019;27(7):990-999. Available from: https://doi.org/10.1016/j.jsps.2019.08.001
- Wang G, Nie Q, Zang C, Zhang B, Zhu Q, Luo G, et al. Self-assembled thermoresponsive nanogels prepared by reverse micelle→positive micelle method for ophthalmic delivery of muscone, a poorly water-soluble drug. Journal of Pharmaceutical Sciences. 2016;105(9):2752-2759. Available from: https://doi.org/10.1016/j.xphs.2016.02.014
- Meng X, Liu J, Yu X, Li J, Lu X, Shen T. Pluronic F127 and D-α-tocopheryl polyethene glycol succinate (TPGS) mixed micelles for targeting drug delivery across the blood-brain barrier. Scientific Reports. 2017;7(1):2964. Available from: https://doi.org/10.1038/s41598-017-03123-y
- Zhuang Y, Yang X, Li Y, Chen Y, Peng X, Yu L, et al. Sustained release strategy designed for lixisenatide delivery to synchronously treat diabetes and associated complications. ACS Applied Materials & Interfaces. 2019;11(33):29604-29618. Available from: https://doi.org/10.1021/acsami.9b10346
- Wang Q, Huang J, Lai Y. Smart drug delivery strategies based on porous nanostructure materials. In: Sezer AD, editor. Smart Drug Delivery System. Rijeka (Croatia): InTech; 2016. p. 63-90. Available from: https://www.intechopen.com/chapters/49794
- Bhawani SA, Nisar M, Tariq A, Alotaibi KM, et al. Enzyme-responsive polymer composites and their applications. In: Smart Polymer Nanocomposites. Elsevier; 2021. p. 169-182. Available from: https://doi.org/10.1016/B978-0-12-819961-9.00017-7
- Gonzalez-Valdivieso J, Girotti A, Schneider J, Arias FJ. Advanced nanomedicine and cancer: Challenges and opportunities in clinical translation. International Journal of Pharmaceutics. 2021;599:120438. Available from: https://doi.org/10.1016/j.ijpharm.2021.120438
- Shahriari M, Zahiri M, Abnous K, Taghdisi SM, Ramezani M, Alibolandi M. Enzyme-responsive drug delivery systems in cancer treatment. Journal of Controlled Release. 2019;308:172-189. Available from: https://doi.org/10.1016/j.jconrel.2019.07.004
- Guo F, Wu J, Wu W, Huang D, Yan Q, Yang Q, et al. PEGylated self-assembled enzyme-responsive nanoparticles for effective targeted therapy against lung tumours. Journal of Nanobiotechnology. 2018;16(1):1-13. Available from: https://doi.org/10.1186/s12951-018-0384-8
- Sun IC, Yoon HY, Lim DK, Kim K. Recent trends in in situ enzyme-activatable prodrugs for targeted cancer therapy. Bioconjugate Chemistry. 2020;31(4):1012-1024. Available from: https://doi.org/10.1021/acs.bioconjchem.0c00082
- Asanuma D, Sakabe M, Kamiya M, Yamamoto K, Hiratake J, Ogawa M, et al. Sensitive β-galactosidase-targeting fluorescence probe for visualising small peritoneal metastatic tumours in vivo. Nature Communications. 2015;6(1):6463. Available from: https://doi.org/10.1038/ncomms7463
- Su FY, Srinivasan S, Lee B, Chen J, Convertine AJ, West TE, Ratner DM, et al. Macrophage-targeted drugamers with enzyme-cleavable linkers deliver high intracellular drug dosing and sustained drug pharmacokinetics against alveolar pulmonary infections. Journal of Controlled Release. 2018;287:1-11. Available from: https://doi.org/10.1016/j.jconrel.2018.08.014
- Awino JK, Gudipati S, Hartmann AK, Santiana JJ, Cairns-Gibson DF, Gomez N, et al. Nucleic acid nanocapsules for enzyme-triggered drug release. Journal of the American Chemical Society. 2017;139(18):6278-6281. Available from: https://doi.org/10.1021/jacs.6b13087
- Zuo J, Tong L, Du L, Yang M, Jin Y. Biomimetic nanoassemblies of 1-O-octodecyl-2-conjugated linoleoyl-sn-glycero-3-phosphatidyl gemcitabine with phospholipase A2-triggered degradation for the treatment of cancer. Colloids and Surfaces B: Biointerfaces. 2017;152:467-474. Available from: https://doi.org/10.1016/j.colsurfb.2017.02.001
- Sharipov M, Tawfik SM, Gerelkhuu Z, Huy BT, Lee YI. Phospholipase A2-responsive phosphate micelle-loaded UCNPs for bioimaging of prostate cancer cells. Scientific Reports. 2017;7(1):16073. Available from: https://doi.org/10.1038/s41598-017-16136-4
- Åhlén M, Tummala GK, Mihranyan A. Nanoparticle-loaded hydrogels as a pathway for enzyme-triggered drug release in ophthalmic applications. International Journal of Pharmaceutics. 2018;536(1):73-81. Available from: https://doi.org/10.1016/j.ijpharm.2017.11.053
- Sur S, Rathore A, Dave V, Reddy KR, Chouhan RS, Sadhu V. Recent developments in functionalized polymer nanoparticles for efficient drug delivery systems. Nano-Structures & Nano-Objects. 2019;20:100397. Available from: https://doi.org/10.1016/j.nanoso.2019.100397
- Jhaveri J, Raichura Z, Khan T, Momin M, Omri A. Chitosan nanoparticles-insight into properties, functionalization and applications in drug delivery and theranostics. Molecules. 2021;26(2):272. Available from: https://doi.org/10.3390/molecules26020272
- Kandasamy G, Maity D. Multifunctional theranostic nanoparticles for biomedical cancer treatments-A comprehensive review. Materials Science and Engineering: C. 2021;127:112199. Available from: https://doi.org/10.1016/j.msec.2021.112199
- Dumontel B, Conejo-Rodríguez V, Vallet-Regí M, Manzano M. Natural biopolymers as smart coating materials of mesoporous silica nanoparticles for drug delivery. Pharmaceutics. 2023;15(2):447. Available from: https://doi.org/10.3390/pharmaceutics15020447
- Amina SJ, Guo B. A review on the synthesis and functionalization of gold nanoparticles as a drug delivery vehicle. International Journal of Nanomedicine. 2020;15:9823-9857. Available from: https://doi.org/10.2147/IJN.S279094
- Yusuf A, Almotairy ARZ, Henidi H, Alshehri OY, Aldughaim MS. Nanoparticles as drug delivery systems: A review of the implication of nanoparticles’ physicochemical properties on responses in biological systems. Polymers. 2023;15(7):1596. Available from: https://doi.org/10.3390/polym15071596
- Ajith S, Almomani F, Elhissi A, Husseini GA. Nanoparticle-based materials in anticancer drug delivery: Current and prospects. Heliyon. 2023;9(11):e21227. Available from: https://doi.org/10.1016/j.heliyon.2023.e21227
- Prieložná J, Mikušová V, Mikuš P. Advances in the delivery of anticancer drugs by nanoparticles and chitosan-based nanoparticles. International Journal of Pharmaceutics: X. 2024;8:100281. Available from: https://doi.org/10.1016/j.ijpx.2024.100281
- Umar AK, Limpikirati PK, Rivai B, Ardiansah I, Sriwidodo S, Luckanagul JA. Complexed hyaluronic acid-based nanoparticles in cancer therapy and diagnosis: Research trends by natural language processing. Heliyon. 2025;11(1):e41246. Available from: https://doi.org/10.1016/j.heliyon.2024.e41246
- Tran PH, Duan W, Tran TT. Fucoidan-based nanostructures: A focus on its combination with chitosan and the surface functionalization of metallic nanoparticles for drug delivery. International Journal of Pharmaceutics. 2020;575:118956. Available from: https://doi.org/10.1016/j.ijpharm.2019.118956
- Huang J, Li Y, Orza A, Lu Q, Guo P, Wang L, et al. Magnetic nanoparticle facilitated drug delivery for cancer therapy with targeted and image-guided approaches. Advanced Functional Materials. 2016;26(22):3818-3836. Available from: https://doi.org/10.1002/adfm.201504185
- Anderson SD, Gwenin VV, Gwenin CD. Magnetic functionalized nanoparticles for biomedical, drug delivery and imaging applications. Nanoscale Research Letters. 2019;14(1):1-16. Available from: https://doi.org/10.1186/s11671-019-3019-6
- Ullah R, Siraj M, Zarshan F, Abbasi BA, Yaseen T, Waris A, et al. A comprehensive overview of fabrication of biogenic multifunctional metal/metal oxide nanoparticles and applications. Reviews in Inorganic Chemistry. 2024;44(0).
- Patra JK, Das G, Fraceto LF, Campos EVR, Rodriguez-Torres MDP, et al. Nano-based drug delivery systems: Recent developments and prospects. Journal of Nanobiotechnology. 2018;16(1):1-33. Available from: https://doi.org/10.1186/s12951-018-0392-8
- MacRitchie N, Frleta-Gilchrist M, Sugiyama A, Lawton T, McInnes IB, Maffia P, et al. Molecular imaging of inflammation - Current and emerging technologies for diagnosis and treatment. Pharmacology & Therapeutics. 2020;211:107550. Available from: https://doi.org/10.1016/j.pharmthera.2020.107550
- Debbage P, Jaschke W. Molecular imaging with nanoparticles: Giant roles for dwarf actors. Histochemistry and Cell Biology. 2008;130(5):845-875. Available from: https://doi.org/10.1007/s00418-008-0511-y
- Rani R, Sethi K, Singh G. Nanomaterials and their applications in bioimaging. In: Prasad R, editor. Plant Nanobionics: Volume 2, Approaches in Nanoparticles, Biosynthesis, and Toxicity. Cham: Springer International Publishing; 2019. p. 429-450. Available from: https://doi.org/10.1007/978-3-030-16379-2_15
- Tian X, Liu S, Zhu J, Qian Z, Bai L, Pan Y, et al. Biofunctional magnetic hybrid nanomaterials for theranostic applications. Nanotechnology. 2018;30(3):032002. Available from: https://doi.org/10.1088/1361-6528/aaebcc
- Luby BM, Charron DM, MacLaughlin CM, Zheng G. Activatable fluorescence: From small molecule to nanoparticle. Advanced Drug Delivery Reviews. 2017;113:97-121. Available from: https://doi.org/10.1016/j.addr.2016.08.010
- Han X, Xu K, Taratula O, Farsad K. Applications of nanoparticles in biomedical imaging. Nanoscale. 2019;11(3):799-819. Available from: https://doi.org/10.1039/c8nr07769j
- Zhao J, Chen J, Ma S, Liu Q, Huang L, Chen X, et al. Recent developments in multimodality fluorescence imaging probes. Acta Pharmaceutica Sinica B. 2018;8(3):320-338. Available from: https://doi.org/10.1016/j.apsb.2018.03.010
- Taghipour YD, Zarebkohan A, Salehi R, Rahimi F, Torchilin VP, Hamblin MR, et al. An update on dual targeting strategy for cancer treatment. Journal of Controlled Release. 2022;349:67-96. Available from: https://doi.org/10.1016/j.jconrel.2022.06.044
- Kosmides AK, Sidhom JW, Fraser A, Bessell CA, Schneck JP. Dual targeting nanoparticle stimulates the immune system to inhibit tumour growth. ACS Nano. 2017;11(6):5417-5429. Available from: https://doi.org/10.1021/acsnano.6b08152
- Stefanick JF, Omstead DT, Kiziltepe T, Bilgicer B. Dual-receptor targeted strategy in nanoparticle design achieves tumour cell selectivity through cooperativity. Nanoscale. 2019;11(10):4414-4427. Available from: https://pubs.rsc.org/en/content/articlelanding/2019/nr/c8nr09431d
- Xia QS, Ding HM, Ma YQ. Can dual-ligand targeting enhance cellular uptake of nanoparticles? Nanoscale. 2017;9(26):8982-8989. Available from: https://pubs.rsc.org/en/content/articlelanding/2017/nr/c7nr01020f
- Vaughan HJ, Green JJ, Tzeng SY. Cancer-targeting nanoparticles for combinatorial nucleic acid delivery. Advanced Materials. 2020;32(13):1901081. Available from: https://doi.org/10.1002/adma.201901081
- Wang S, Huang P, Chen X. Hierarchical targeting strategy for enhanced tumour tissue accumulation/retention and cellular internalisation. Advanced Materials. 2016;28(34):7340-7364. Available from: https://doi.org/10.1002/adma.201601498
- Gatti THH, Eloy JO, Ferreira LMB, da Silva IC, Pavan FR, Gremião MPD, et al. Insulin-loaded polymeric mucoadhesive nanoparticles: Development, characterisation, and cytotoxicity evaluation. Brazilian Journal of Pharmaceutical Sciences. 2018;54. Available from: https://doi.org/10.1590/s2175-97902018000117314
- Chenthamara D, Subramaniam S, Ramakrishnan SG, Krishnaswamy S, Essa MM, et al. Therapeutic efficacy of nanoparticles and routes of administration. Biomaterials Research. 2019;23(1):1-29. Available from: https://doi.org/10.1186/s40824-019-0166-x
- de Almeida MS, Susnik E, Drasler B, Taladriz-Blanco P, Petri-Fink A, Rothen-Rutishauser B. Understanding nanoparticle endocytosis to improve targeting strategies in nanomedicine. Chemical Society Reviews. 2021;50(9):5397-5434. Available from: https://doi.org/10.1039/d0cs01127d
- Zhao MX, Zhu BJ. The research and applications of quantum dots as nano-carriers for targeted drug delivery and cancer therapy. Nanoscale Research Letters. 2016;11:1-9. Available from: https://doi.org/10.1186/s11671-016-1394-9
- Khurana A, Tekula S, Saifi MA, Venkatesh P, Godugu C. Therapeutic applications of selenium nanoparticles. Biomedicine & Pharmacotherapy. 2019;111:802-812. Available from: https://doi.org/10.1016/j.biopha.2018.12.146
- Parodi A, Molinaro R, Sushnitha M, Evangelopoulos M, Martinez JO, Arrighetti N, et al. Bio-inspired engineering of cell- and virus-like nanoparticles for drug delivery. Biomaterials. 2017;147:155-168. Available from: https://doi.org/10.1016/j.biomaterials.2017.09.020
- Ly PD, Ly KN, Phan HL, Nguyen HHT, Duong VA, Nguyen HV, et al. Recent advances in surface decoration of nanoparticles in drug delivery. Frontiers in Nanotechnology. 2024;6. Available from: https://doi.org/10.3389/fnano.2024.1456939
- Liang YJ, Xie J, Yu J, Zheng Z, Liu F, Yang A, et al. Recent advances of high-performance magnetic iron oxide nanoparticles: Controlled synthesis, properties tuning and cancer theranostics. Nano Select. 2021;2(2):216-250. Available from: https://doi.org/10.1002/nano.202000169
- Ansari AA, Parchur AK, Thorat ND, Chen G. New advances in pre-clinical diagnostic imaging perspectives of functionalized upconversion nanoparticle-based nanomedicine. Coordination Chemistry Reviews. 2021;440:213971. Available from: https://doi.org/10.1016/j.ccr.2021.213971
- Kravanja KA, Finšgar M. A review of techniques for the application of bioactive coatings on metal-based implants to achieve controlled release of active ingredients. Materials & Design. 2022;217:110653. Available from: https://doi.org/10.1016/j.matdes.2022.110653
- Dadfar SM, Roemhild K, Drude NI, von Stillfried S, Knüchel R, Kiessling F, et al. Iron oxide nanoparticles: Diagnostic, therapeutic and theranostic applications. Advanced Drug Delivery Reviews. 2019;138:302-325. Available from: https://doi.org/10.1016/j.addr.2019.01.005
- Komsthöft T, Bovone G, Bernhard S, Tibbitt MW, et al. Polymer functionalization of inorganic nanoparticles for biomedical applications. Current Opinion in Chemical Engineering. 2022;37:100849. Available from: https://doi.org/10.1016/j.coche.2022.100849
- Wang W, Mattoussi H. Engineering the bio–nano interface using a multifunctional coordinating polymer coating. Accounts of Chemical Research. 2020;53(6):1124-1138. Available from: https://doi.org/10.1021/acs.accounts.9b00641
- Sanchez-Cano C, Carril M. Recent developments in the design of non-biofouling coatings for nanoparticles and surfaces. International Journal of Molecular Sciences. 2020;21(3):1007. Available from: https://doi.org/10.3390/ijms21031007
- Zou Y, Ito S, Yoshino F, Suzuki Y, Zhao L, Komatsu K. Polyglycerol grafting shields nanoparticles from protein corona formation to avoid macrophage uptake. ACS Nano. 2020;14(6):7216-7226. Available from: https://doi.org/10.1021/acsnano.0c02289
- Maan AM, Hofman AH, de Vos WM, Kamperman M. Recent developments and practical feasibility of polymer-based antifouling coatings. Advanced Functional Materials. 2020;30(32):2000936. Available from: https://doi.org/10.1002/adfm.202000936
- Zhang P, Ratner BD, Hoffman AS, Jiang S. Nonfouling surfaces. In: Biomaterials Science. Elsevier; 2020. p. 507-513. Available from: https://doi.org/10.1016/B978-0-12-816137-1.00034-9
- Zheng L, Sundaram HS, Wei Z, Li C, Yuan Z. Applications of zwitterionic polymers. Reactive and Functional Polymers. 2017;118:51-61. Available from: https://doi.org/10.1016/j.reactfunctpolym.2017.07.006
- Kozma GT, Shimizu T, Ishida T, Szebeni J. Anti-PEG antibodies: Properties, formation, testing and role in adverse immune reactions to PEGylated nano-biopharmaceuticals. Advanced Drug Delivery Reviews. 2020;154:163-175. Available from: https://doi.org/10.1016/j.addr.2020.07.024
- Aghajani M, Esmaeili F. Anti-biofouling assembly strategies for protein & cell repellent surfaces: A mini-review. Journal of Biomaterials Science, Polymer Edition. 2021;32(13):1770-1789. Available from: https://doi.org/10.1080/09205063.2021.1932357
- Debayle M, Balloul E, Dembele F, Xu X, Hanafi M, Ribot F, et al. Zwitterionic polymer ligands: An ideal surface coating to totally suppress protein-nanoparticle corona formation? Biomaterials. 2019;219:119357. Available from: https://doi.org/10.1016/j.biomaterials.2019.119357
- Li W, Chu K, Liu L. Zwitterionic gel coating endows gold nanoparticles with ultrastability. Langmuir. 2018;35(5):1369-1378. Available from: https://doi.org/10.1021/acs.langmuir.8b01600
- Morgese G, Benetti EM. Polyoxazoline biointerfaces by surface grafting. European Polymer Journal. 2017;88:470-485. Available from: https://doi.org/10.1016/j.eurpolymj.2016.11.003
- Giner-Casares JJ, Henriksen-Lacey M, Coronado-Puchau M, Liz-Marzán LM. Inorganic nanoparticles for biomedicine: Where materials scientists meet medical research. Materials Today. 2016;19(1):19-28. Available from: https://doi.org/10.1016/j.mattod.2015.07.004
- Simons K. Cell membranes: A subjective perspective. Biochimica et Biophysica Acta (BBA)-Biomembranes. 2016;1858(10):2569-2572. Available from: https://doi.org/10.1016/j.bbamem.2016.01.023
- Guan X, Xing S, Liu Y. Engineered cell membrane-camouflaged nanomaterials for biomedical applications. Nanomaterials. 2024;14(5):413. Available from: https://doi.org/10.3390/nano14050413
- Jiao M, Zhang P, Meng J, Li Y, Liu C, Luo X, Gao M. Recent advancements in biocompatible inorganic nanoparticles towards biomedical applications. Biomaterials Science. 2018;6(4):726-745. Available from: https://doi.org/10.1039/c7bm01020f
- Campora S, Ghersi G. Recent developments and applications of smart nanoparticles in biomedicine. Nanotechnology Reviews. 2022;11(1):2595-2631. Available from: https://doi.org/10.1515/ntrev-2022-0148
- Luchini A, Vitiello G. Understanding the nano-bio interfaces: Lipid-coatings for inorganic nanoparticles as a promising strategy for biomedical applications. Frontiers in Chemistry. 2019;7:343. Available from: https://doi.org/10.3389/fchem.2019.00343
- Park YI, Kim E, Huang CH, Park KS, Castro CM, Lee H, et al. Facile coating strategy to functionalize inorganic nanoparticles for biosensing. Bioconjugate Chemistry. 2017;28(1):33-37. Available from: https://doi.org/10.1021/acs.bioconjchem.6b00524
- Harayama T, Riezman H. Understanding the diversity of membrane lipid composition. Nature Reviews Molecular Cell Biology. 2018;19(5):281-296. Available from: https://doi.org/10.1038/nrm.2017.138
- Disalvo A, Frias MA. Surface characterisation of lipid biomimetic systems. Membranes. 2021;11(11):821. Available from: https://doi.org/10.3390/membranes11110821
- Waghule T, Saha RN, Alexander A, Singhvi G. Tailoring the multi-functional properties of phospholipids for simple to complex self-assemblies. Journal of Controlled Release. 2022;349:460-474. Available from: https://doi.org/10.1016/j.jconrel.2022.07.014
- Suk JS, Xu Q, Kim N, Hanes J, Ensign LM. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Advanced Drug Delivery Reviews. 2016;99:28-51. Available from: https://doi.org/10.1016/j.addr.2015.09.012
- Shah S, Rangaraj N, Singh SB, Srivastava S. Exploring the unexplored avenues of surface charge in nano-medicine. Colloid and Interface Science Communications. 2021;42:100406. Available from: https://doi.org/10.1016/j.colcom.2021.100406
- Mirahadi M, Ghanbarzadeh S, Ghorbani M, Gholizadeh A, Hamishehkar H. A review on the role of lipid-based nanoparticles in medical diagnosis and imaging. Therapeutic Delivery. 2018;9(8):557-569. Available from: https://doi.org/10.4155/tde-2018-0020
- Pavel IA, Girardon M, El Hajj S, Parant S, Amadei F, Kaufmann S, et al. Lipid-coated mesoporous silica microparticles for the controlled delivery of β-galactosidase into intestines. Journal of Materials Chemistry B. 2018;6(35):5633-5639. Available from: https://pubs.rsc.org/en/content/articlelanding/2018/tb/c8tb01114a
- Gonzalez-Pech N, Grassian V. Surface chemical functionalities of environmental nanomaterials. 2018. Available from: https://www.researchgate.net/publication/321077841_Surface_Chemical_Functionalities_of_Environmental_Nanomaterials
- Gong YN, Zhu B, Bu YZ, Du BJ, Liu SC, Luo L, et al. Thiolated nanomaterials for bone tissue engineering: Synthesis, mechanisms, and applications. Rare Metals. 2025:1-30. Available from: https://doi.org/10.1007/s12598-024-03212-8
- Lu L, Duong VT, Shalash AO, Skwarczynski M, Toth I. Chemical conjugation strategies for the development of protein-based subunit nanovaccines. Vaccines. 2021;9(6):563. Available from: https://doi.org/10.3390/vaccines9060563
- Tsuchikama K, An Z. Antibody-drug conjugates: Recent advances in conjugation and linker chemistries. Protein & Cell. 2018;9(1):33-46. Available from: https://doi.org/10.1007/s13238-016-0323-0
- Yanase N, Toyota H, Hata K, Yagyu S, Seki T, Harada M, et al. OVA-bound nanoparticles induce OVA-specific IgG1, IgG2a, and IgG2b responses with low IgE synthesis. Vaccine. 2014;32(45):5918-5924. Available from: https://doi.org/10.1016/j.vaccine.2014.08.059
- Sanchez-Villamil J, Tapia D, Torres A. Development of a gold nanoparticle vaccine against enterohemorrhagic Escherichia coli O157:H7. mBio. 2019;10:e01869-19. Available from: https://doi.org/10.1128/mbio.01869-19
- Faruck MO, Zhao L, Hussein WM, Khalil ZG, Capon RJ, Skwarczynski M, et al. Polyacrylate–peptide antigen conjugate as a single-dose oral vaccine against Group A Streptococcus. Vaccines. 2020;8(1):23. Available from: https://doi.org/10.3390/vaccines8010023
- Chen J, Wang J, Li K, Wang Y, Gruebele M, Ferguson AL, et al. Polymeric “clickase” accelerates the copper click reaction of small molecules, proteins, and cells. Journal of the American Chemical Society. 2019;141(24):9693-9700. Available from: https://pubs.acs.org/doi/10.1021/jacs.9b04181
- Pickens CJ, Johnson SN, Pressnall MM, Leon MA, Berkland CJ. Practical considerations, challenges, and limitations of bioconjugation via azide–alkyne cycloaddition. Bioconjugate Chemistry. 2017;29(3):686-701. Available from: https://pubs.acs.org/doi/10.1021/acs.bioconjchem.7b00633
- Greene MK, Richards DA, Nogueira JCF, Campbell K, Smyth P, Fernández M, et al. Forming next-generation antibody–nanoparticle conjugates through the oriented installation of non-engineered antibody fragments. Chemical Science. 2018;9(1):79-87. Available from: https://doi.org/10.1039/c7sc02747h
- Song F, Chan WC. Principles of conjugating quantum dots to proteins via carbodiimide chemistry. Nanotechnology. 2011;22(49):494006. Available from: https://doi.org/10.1088/0957-4484/22/49/494006
- Shinde P, Kumar A, Kavitha, Dey K, Mohan L, Kar S, et al. Physical approaches for drug delivery: An overview. In: Delivery of Drugs. 2020:161-190. Available from: https://research.uees.edu.ec/en/publications/physical-approaches-for-drug-delivery-an-overview-an-overview-2/
- Moreira-Alvarez B, Cid-Barrio L, Ferreira HS, Costa-Fernández JM, Ruiz Encinar J. Integrated analytical platforms for the comprehensive characterisation of bioconjugated inorganic nanomaterials aiming at biological applications. Journal of Analytical Atomic Spectrometry. 2020;35(8):1518-1529. Available from: https://pubs.rsc.org/en/content/articlelanding/2020/ja/d0ja00147c
- Zhang W, Taheri-Ledari R, Ganjali F, Afruzi FH, Hajizadeh Z, Saeidirad M, et al. Nanoscale bioconjugates: A review of the structural attributes of drug-loaded nanocarrier conjugates for selective cancer therapy. Heliyon. 2022;8(6):e09577. Available from: https://doi.org/10.1016/j.heliyon.2022.e09577
- Awan M, Rauf S, Abbas A, Nawaz MH, Yang C, Shahid SA, et al. A sandwich electrochemical immunosensor based on antibody functionalized-silver nanoparticles (Ab-Ag NPs) for the detection of dengue biomarker protein NS1. Journal of Molecular Liquids. 2020;317:114014. Available from: https://doi.org/10.1016/j.molliq.2020.114014
- Chakraborty S, Dlie ZY, Chakraborty S, Roy S, Mukherjee B, Besra SE, et al. Aptamer-functionalized drug nanocarrier improves hepatocellular carcinoma toward normal by targeting neoplastic hepatocytes. Molecular Therapy-Nucleic Acids. 2020;20:34-49. Available from: https://doi.org/10.1016/j.omtn.2020.01.034
- Chen Y, Deng Y, Zhu C, Xiang C. Anti-prostate cancer therapy: Aptamer-functionalized, curcumin and cabazitaxel co-delivered, tumour targeted lipid-polymer hybrid nanoparticles. Biomedicine & Pharmacotherapy. 2020;127:110181. Available from: https://doi.org/10.1016/j.biopha.2020.110181
- Gessner I, Klimpel A, Klußmann M, Neundorf I, Mathur S. Interdependence of charge and secondary structure on cellular uptake of cell-penetrating peptide functionalized silica nanoparticles. Nanoscale Advances. 2020;2(1):453-462. Available from: https://doi.org/10.1039/c9na00693a
- Sampogna-Mireles D, Araya-Durán ID, Márquez-Miranda V, Valencia-Gallegos JA, González-Nilo FD. Structural analysis of binding functionality of folic acid-PEG dendrimers against folate receptor. Journal of Molecular Graphics and Modelling. 2017;72:201-208. Available from: https://doi.org/10.1016/j.jmgm.2017.01.004
- Li Y, Wang S, Song FX, Zhang L, Yang W, Wang HX, et al. A pH-sensitive drug delivery system based on folic acid-targeted HBP-modified mesoporous silica nanoparticles for cancer therapy. Colloids and Surfaces A: Physicochemical and Engineering Aspects. 2020;590:124470. Available from: https://doi.org/10.1016/j.colsurfa.2020.124470
- Taheri-Ledari R, Fazeli A, Kashtiaray A, Salek Soltani S, Maleki A, Zhang W. Cefixime-containing silica nanoseeds coated by a hybrid PVA-gold network with a Cys–Arg dipeptide conjugation: Enhanced antimicrobial and drug release properties. Langmuir. 2021;38(1):132-146. Available from: https://doi.org/10.1021/acs.langmuir.1c02233
- Zhang W, Taheri-Ledari R, Ganjali F, Afruzi FH, Hajizadeh Z, Saeidirad M, et al. Nanoscale bioconjugates: A review of the structural attributes of drug-loaded nanocarrier conjugates for selective cancer therapy. Heliyon. 2022;8(6):e09577. Available from: https://doi.org/10.1016/j.heliyon.2022.e09577
- Cai H, Liang Z, Huang W, Wen L, Chen G. Engineering PLGA nano-based systems through understanding the influence of nanoparticle properties and cell-penetrating peptides for cochlear drug delivery. International Journal of Pharmaceutics. 2017;532(1):55-65. Available from: https://doi.org/10.1016/j.ijpharm.2017.08.084
- Conte C, Longobardi G, Barbieri A, Palma G, Luciano A, Dal Poggetto G, et al. Non-covalent strategies to functionalize polymeric nanoparticles with NGR peptides for targeting breast cancer. International Journal of Pharmaceutics. 2023;633:122618. Available from: https://doi.org/10.1016/j.ijpharm.2023.122618
- Jha R, Mayanovic RA. A review of the preparation, characterisation, and applications of chitosan nanoparticles in nanomedicine. Nanomaterials. 2023;13(8):1302. Available from: https://doi.org/10.3390/nano13081302
- Vashi A. Innovative approaches in characterising and developing methods for lipoidal vesicular drug delivery systems. GSC Advanced Research and Reviews. 2024;20(1):427-438. Available from: https://doi.org/10.30574/gscarr.2024.20.1.0278
- Jia Z, Li J, Gao L, Yang D, Kanaev A. Dynamic light scattering: A powerful tool for in situ nanoparticle sizing. Colloids and Interfaces. 2023;7(1):15. Available from: https://www.mdpi.com/2504-5377/7/1/15
- Kowkabany G, Bao Y. Nanoparticle tracking analysis: An effective tool to characterise extracellular vesicles. Molecules. 2024;29(19):4672. Available from: https://doi.org/10.3390/molecules29194672
- Lin Y, Zhou M, Tai X, Li H, Han X, Yu J. Analytical transmission electron microscopy for emerging advanced materials. Matter. 2021;4(7):2309-2339. Available from: https://doi.org/10.1016/j.matt.2021.05.005
- Ophus C. Quantitative scanning transmission electron microscopy for materials science: Imaging, diffraction, spectroscopy, and tomography. Annual Review of Materials Research. 2023;53(1):105-141. Available from: https://doi.org/10.1146/annurev-matsci-080921-092646
- Matatyaho Ya’akobi A, Talmon Y. Extending cryo-EM to nonaqueous liquid systems. Accounts of Chemical Research. 2021;54(9):2100-2109. Available from: https://doi.org/10.1021/acs.accounts.1c00077
- Liu Q, Fu Y, Qin Z, Wang Y, Zhang S, Ran M. Progress in the applications of atomic force microscope (AFM) for mineralogical research. Micron. 2023;170:103460. Available from: https://doi.org/10.1016/j.micron.2023.103460
- Adelantado C, Jordens J, Voorspoels S, Velimirovic M, Tirez K. Assessment of electrophoretic mobility determination in nanoparticle analysis: Two parallel techniques converging in a distinctive parameter. Electrophoresis. 2025:e202400132. Available from: https://doi.org/10.1002/elps.202400132
- Heidari Z, Salehzadeh A, Sadat Shandiz SA, Tajdoost S. Anti-cancer and anti-oxidant properties of ethanolic leaf extract of Thymus vulgaris and its bio-functionalized silver nanoparticles. 3 Biotech. 2018;8(3):1-14. Available from: https://doi.org/10.1007/s13205-018-1199-x
- Lambadi PR, Sharma TK, Kumar P, Vasnani P, Thalluri SM, Bisht N, et al. Facile biofunctionalisation of silver nanoparticles for enhanced antibacterial properties, endotoxin removal, and biofilm control. International Journal of Nanomedicine. 2015;10:2155-2171. Available from: https://doi.org/10.2147/IJN.S72923
- Kumar VV, Anbarasan S, Christena LR, SaiSubramanian N, Philip Anthony S. Bio-functionalized silver nanoparticles for selective colourimetric sensing of toxic metal ions and antimicrobial studies. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 2014;129:35-42. Available from: https://doi.org/10.1016/j.saa.2014.03.020
- Kalam A, Al-Sehemi AG, Alrumman S, Du G, Assiri M, Hesham AEL. Antibacterial studies of bio-functionalized carbon decorated silver nanoparticles (AgNPs). Journal of the Indian Chemical Society. 2021;98(10):100155. Available from: https://doi.org/10.1016/j.jics.2021.100155
- Sarkar K, Banerjee SL, Kundu PP, Madras G, Chatterjee K. Biofunctionalized surface-modified silver nanoparticles for gene delivery. Journal of Materials Chemistry B. 2015;3(26):5266-5276. Available from: https://doi.org/10.1039/c5tb00614g
- Dong Y, Hong M, Dai R, Wu H, Zhu P. Engineered bioactive nanoparticles incorporated biofunctionalized ECM/silk proteins based cardiac patches combined with MSCs for the repair of myocardial infarction: In vitro and in vivo evaluations. Science of the Total Environment. 2020;707:135976. Available from: https://doi.org/10.1016/j.scitotenv.2019.135976
- Vojtech JM, Cano-Mejia J, Dumont MF, Sze RW, Fernandes R. Biofunctionalized Prussian blue nanoparticles for multimodal molecular imaging applications. Journal of Visualized Experiments. 2015;(98):e52621. Available from: https://doi.org/10.3791/52621
- Zhang P, Ouyang Q, Zhai T, Sun J, Wu J, Qin F, et al. An inflammation-targeted nanoparticle with bacteria-forced release of polymyxin B for pneumonia therapy. Nanoscale. 2022;14(41):15291-15304. Available from: https://doi.org/10.1039/d2nr02026b
- Gounani Z, Asadollahi MA, Meyer RL, Arpanaei A. Loading of polymyxin B onto anionic mesoporous silica nanoparticles retains antibacterial activity and enhances biocompatibility. International Journal of Pharmaceutics. 2018;537(1-2):148-161. Available from: https://doi.org/10.1016/j.ijpharm.2017.12.039
- Chakravarty R, Goel S, Hong H, Chen F, Valdovinos HF, Hernandez R, et al. Hollow mesoporous silica nanoparticles for tumour vasculature targeting and PET image-guided drug delivery. Nanomedicine. 2015;10(8):1233-1246. Available from: https://doi.org/10.2217/nnm.14.226
- Sanità G, Carrese B, Lamberti A. Nanoparticle surface functionalization: How to improve biocompatibility and cellular internalisation. Frontiers in Molecular Biosciences. 2020;7:587012. Available from: https://doi.org/10.3389/fmolb.2020.587012
- Zahiri M, Babaei M, Abnous K, Taghdisi SM, Ramezani M, Alibolandi M. Hybrid nanoreservoirs based on dextran-capped dendritic mesoporous silica nanoparticles for CD133-targeted drug delivery. Journal of Cellular Physiology. 2020;235(2):1036-1050. Available from: https://doi.org/10.1002/jcp.29019
- Chen L, Zhou X, He C. Mesoporous silica nanoparticles for tissue-engineering applications. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology. 2019;11(6):e1573. Available from: https://doi.org/10.1002/wnan.1573
- Zahiri M, Falsafi M, Lamei K, Abnous K, Taghdisi SM, Ramezani M, et al. Targeted biomimetic hollow mesoporous organosilica nanoparticles for delivery of doxorubicin to colon adenocarcinoma: In vitro and in vivo evaluation. Microporous and Mesoporous Materials. 2022;335:111841. Available from: https://doi.org/10.1016/j.micromeso.2022.111841
- Seebacher NA, Richardson DR, Jansson PJ. A mechanism for overcoming P-glycoprotein-mediated drug resistance: Novel combination therapy that releases stored doxorubicin from lysosomes via lysosomal permeabilisation using Dp44mT or DpC. Cell Death & Disease. 2016;7(12):e2510. Available from: https://doi.org/10.1038/cddis.2016.381
- Mirab F, Kang YJ, Majd S. Preparation and characterisation of size-controlled glioma spheroids using agarose hydrogel microwells. PLoS One. 2019;14(1):e0211078. Available from: https://doi.org/10.1371/journal.pone.0211078
- Ge J, Jia Q, Liu W, Lan M, Zhou B, Guo L, et al. Carbon dots with intrinsic theranostic properties for bioimaging, red-light-triggered photodynamic/photothermal simultaneous therapy in vitro and in vivo. Advanced Healthcare Materials. 2016;5(6):665-675. Available from: https://doi.org/10.1002/adhm.201500720
- Ren Y, Zhang Y, Li X. Application of AgNPs in biomedicine: An overview and current trends. Nanotechnology Reviews. 2024;13(1):20240030. Available from: https://doi.org/10.1515/ntrev-2024-0030
- Lynch I, Feitshans IL, Kendall M. ‘Bio-nano interactions: New tools, insights and impacts’: Summary of the Royal Society discussion meeting. Philosophical Transactions of the Royal Society B: Biological Sciences. 2015;370(1661):20140162. Available from: https://doi.org/10.1098/rstb.2014.0162
- Hheidari A, Mohammadi J, Ghodousi M, Mahmoodi M, Ebrahimi S, Pishbin E, et al. Metal-based nanoparticles in cancer treatment: Lessons learned and challenges. Frontiers in Bioengineering and Biotechnology. 2024;12:1436297. Available from: https://doi.org/10.3389/fbioe.2024.1436297
- Ninan N, Albrecht H, Blencowe A. Mammalian cell-based assays for studying bio-nano interactions, in Characterisation of Nanomaterials. Elsevier. 2018;129-166. Available from: https://www.researchgate.net/publication/325940964_Mammalian_Cell-Based_Assays_for_Studying_Bio-Nano_Interactions
- De Simone U, Spinillo A, Caloni F, Avanzini MA, Coccini T. In vitro evaluation of magnetite nanoparticles in human mesenchymal stem cells: Comparison of different cytotoxicity assays. Toxicology Mechanisms and Methods. 2020;30(1):48-59. Available from: https://doi.org/10.1080/15376516.2019.1650151
- Xu Y, Hadjiargyrou M, Rafailovich M, Mironava T. Cell-based cytotoxicity assays for engineered nanomaterials safety screening: Exposure of adipose-derived stromal cells to titanium dioxide nanoparticles. Journal of Nanobiotechnology. 2017;15(1):1-17. Available from: https://doi.org/10.1186/s12951-017-0285-2
- Bailly A-L, Correard F, Popov A, Tselikov G, Chaspoul F, Appay R, et al. In vivo evaluation of safety, biodistribution and pharmacokinetics of laser-synthesised gold nanoparticles. Scientific Reports. 2019;9(1):12890. Available from: https://doi.org/10.1038/s41598-019-48748-3
- Meng F, Wang J, Ping Q, Yeo Y. Quantitative assessment of nanoparticle biodistribution by fluorescence imaging, revisited. ACS Nano. 2018;12(7):6458-6468. Available from: https://doi.org/10.1021/acsnano.8b02881
- Licciardello N, Hunoldt S, Bergmann R, Singh G, Mamat C, Faramus A, et al. Biodistribution studies of ultrasmall silicon nanoparticles and carbon dots in experimental rats and tumour mice. Nanoscale. 2018;10(21):9880-9891. Available from: https://doi.org/10.1039/c8nr01063c
- Meir R, Popovtzer R. Cell tracking using gold nanoparticles and computed tomography imaging. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology. 2018;10(2):e1480. Available from: https://doi.org/10.1002/wnan.1480
- Goodfellow FT, Simchick GA, Mortensen LJ, Stice SL, Zhao Q. Tracking and quantification of magnetically labelled stem cells using magnetic resonance imaging. Advanced Functional Materials. 2016;26(22):3899-3915. Available from: https://doi.org/10.1002/adfm.201504444